
5.5/6.5 Orthopedic Series Spinal System Pedicle Spinal Screws

Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm Spinal Pedicle Screw Instrument Set

Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System Surgical Instrument Set
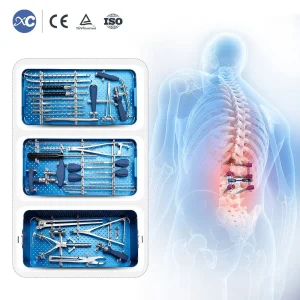
Orthopedic Bone Surgical Set 6.0mm Spinal Pedicle Screw System Instrument Set

Dial-Adjustable Weight Stack System with Chrome Plated Guide Rods

Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set

Orthopedic Spinal Fixation Medical Instrument Vertebral Hook
The global orthopedic implant market has witnessed a paradigm shift toward advanced biomaterials, with Titanium Spinal Rods emerging as the gold standard for posterior spinal fixation. As the medical community tackles an aging population and rising incidences of spinal disorders, the demand for high-strength, biocompatible, and MRI-compatible fixation systems has skyrocketed.
The global spinal implants market is projected to reach billions by 2030, driven by the expansion of healthcare infrastructure in emerging economies across Asia-Pacific and Latin America.
Research is shifting from pure titanium to advanced Grade 5 Titanium alloys (Ti6Al4V), offering superior fatigue resistance and load-bearing capabilities for complex surgeries.
Modern spinal rods are now designed to be compatible with robotic-assisted surgery systems, ensuring sub-millimeter precision in placement and fixation.
Titanium spinal rods are not merely hardware; they are life-altering components that provide stabilization during the critical fusion process. From treating degenerative disc diseases to correcting severe idiopathic scoliosis, these rods must withstand millions of cycles of physiological stress. This industrial reality mandates a manufacturing partner who understands both the mechanical engineering and biological implications of implantable devices.
Immediate availability and sterilized packaging are paramount for emergency spinal trauma cases where every second counts.
Bulk procurement needs often focus on regulatory compliance (CE, ISO, FDA) and consistent batch-to-batch quality to meet national standards.
Customization of rod lengths, diameters (5.5mm/6.0mm), and surface finishes (anodization) allows brands to tailor solutions to specific surgical techniques.
Localized Scenario - Aging Societies: In regions like Europe and East Asia, the rise in osteoporotic spinal fractures has increased the demand for "low-profile" titanium rods that minimize soft tissue irritation while providing maximum rigidity. As a leading exporter, we provide specialized solutions that cater to these demographic-specific surgical needs.
1. Minimally Invasive Surgery (MIS): There is a significant move toward MIS techniques. Suppliers must now provide percutaneous rod insertion tools and compatible rods that reduce incision sizes and accelerate recovery times.
2. Patient-Specific Implants: Using 3D printing (Additive Manufacturing) technology to create patient-matched rod contours is the future. While standard rods remain the backbone of the industry, the integration of 3D anatomical modeling is becoming a key competitive edge.
3. Anti-Microbial Surfaces: The industry is exploring coatings that reduce the risk of implant-associated infections, a major concern for long-term spinal fixation.
Changsha WEX Medical Instruments Co., Ltd. has experiences of more than two decades in areas of innovation and production of orthopaedic implants, such as bone plates, bone screws, interlocking nails, spine implants/internal fixation systems, and all kinds of instruments, which are branded by "WEX". All the implant products have certification of CE and ISO9001/ISO13485, so can reach different requirements of customers.
With a Quality Management System passed ISO 9001:2008 and ISO 13485:2003, we have bright and spacious factory workshops; more than hundred production equipments, such as advanced CNC machining centre machines, CNC sliding headstock auto-lathes, CNC milling machines, CNC lathing machines, upmarket ultrasonic cleaning equipments and etc; more than ten advanced and well-appointed testing equipment, such as Projectors, Tensile-testing machines, Durometers, Carbon and sulphur analyzers, Pitting LP detecting instruments and Fluorescent fault detectors and so on. The packaging process is conducted in clean workshop line with GMP standard.


At the beginning of company's establishment, we aim to design and produce implantable products professionally for our customers. Relying on the operational systems of development, design, production and marketing, which is formed by the orthopaedic experts in domestic large and medium-sized hospitals, also we completed the clinical insurance for our products.
We satisfy our customer's requirements with best quality and best service, based on our advanced production and processing technology. Because we insist on the idea of "Be professional for manufacturing high-quality products, be wholehearted for serving customers."
When selecting a Titanium Spinal Rods supplier, technical specifications are the cornerstone of trust. Our rods are manufactured using medical-grade Titanium Alloy (Ti6Al4V ELI), which provides an optimal balance between biocompatibility and mechanical strength. The Elastic Modulus of titanium is closer to human bone compared to stainless steel, which significantly reduces the "stress shielding" effect, promoting better natural bone healing.
Our Spinal Pedicle Screw Systems are designed with a 5.5mm or 6.0mm diameter architecture, ensuring compatibility with standard surgical instrumentation globally. Each rod undergoes rigorous surface treatment processes. We utilize anodic oxidation to create a biologically inert surface that enhances corrosion resistance and provides color-coding for different sizes, aiding surgeons in the operating room environment.
Quality control at WEX is not just a department; it is our culture. From raw material inspection using carbon and sulphur analyzers to final product testing on tensile-testing machines, we ensure every rod can withstand the dynamic loads of the human spine. Our cleanroom facilities follow stringent GMP standards, ensuring that every implant reaches the surgeon in a state of pristine sterility, ready for life-saving procedures.
As a leading exporter from China, we understand the logistical challenges of international medical trade. Our supply chain is optimized for rapid response, offering flexible lead times and robust packaging that maintains integrity across transcontinental shipping. Whether you are a large-scale hospital group in the Middle East or a specialized orthopedic distributor in Southeast Asia, our team provides the technical documentation and regulatory support needed to streamline your procurement process.