Advanced Orthopedic Engineering | Global Standards | Precision Manufacturing
The global orthopedic implant market, particularly for intramedullary nails, is witnessing rapid transformation driven by an aging global population and the increasing prevalence of trauma-related injuries. Intramedullary nailing, as a minimally invasive gold standard for fracture fixation, has become a focus for innovation. Currently, the industry is shifting towards advanced titanium alloys, biocompatible coatings, and modular designs that accommodate different anatomical variations.
In the industrial landscape, manufacturers are increasingly adopting AI-driven CNC precision machining to ensure that the "lock" of the intramedullary nail is secure and that the fatigue resistance meets stringent international standards. As a result, healthcare procurement departments in Europe, North America, and emerging markets are prioritizing suppliers who demonstrate not only cost-effectiveness but also deep technical competence in material science and regulatory adherence.
China has emerged as the global epicenter for medical device manufacturing, combining high-tech production capabilities with a robust, integrated supply chain. For international distributors and hospitals, partnering with a top-tier Chinese factory offers several strategic advantages:
Changsha WEX Medical Instruments Co., Ltd. brings more than two decades of dedicated experience to the innovation and production of orthopedic implants. Our portfolio, branded as "WEX", encompasses bone plates, bone screws, interlocking nails, and comprehensive internal fixation systems. We are not just manufacturers; we are partners in patient recovery.
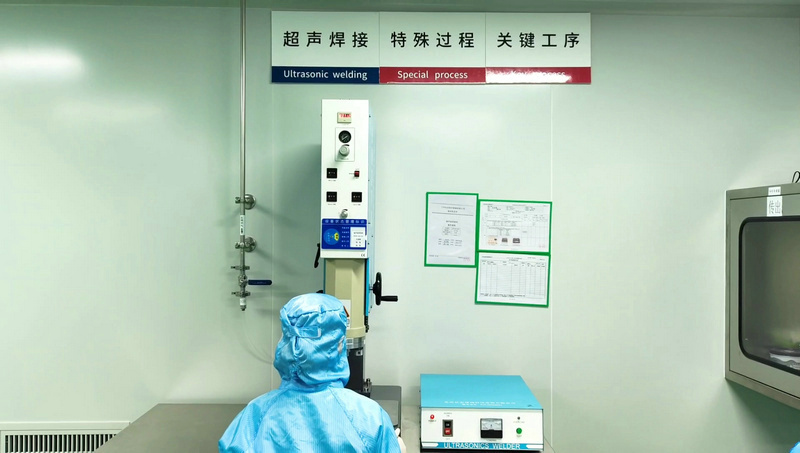
With a Quality Management System certified under ISO 9001:2008 and ISO 13485:2003, our facilities are equipped with the latest in manufacturing technology, including CNC machining centers, auto-lathes, and ultrasonic cleaning systems. Our clean workshop lines are built strictly to GMP standards, ensuring that every product—from an interlocking nail to a spinal implant—is sterile and safe for clinical use.
Our commitment goes beyond production. We operate on a cycle of development, design, and marketing formed by the collaboration with orthopedic experts in major domestic hospitals. This clinical integration allows us to refine our product designs based on real-world surgical feedback, ensuring our tools perform optimally in high-stakes operating room environments. Our philosophy is simple: "Be professional for manufacturing high-quality products, be wholehearted for serving customers."