The global intramedullary (IM) nails market is currently experiencing a transformative phase, driven by the increasing incidence of orthopedic injuries resulting from road accidents and a growing geriatric population prone to osteoporotic fractures. As a specialized Intramedullary Nails Factory & Exporter, we recognize that IM nailing has become the "Gold Standard" for treating long bone fractures in the femur, tibia, and humerus.
Technological advancements in the design of internal fixation systems have significantly improved patient outcomes. Modern interlocking nails offer better rotational stability and load-sharing capabilities, which promote earlier mobilization and faster healing. The shift towards minimally invasive surgery (MIS) has further boosted the demand for high-precision intramedullary systems that can be inserted through small incisions, reducing surgical trauma and hospital stay duration.

Integration of sensor technology to monitor bone healing and load distribution in real-time, providing surgeons with critical post-operative data.
Development of nails with hydroxyapatite or antimicrobial coatings to enhance osseointegration and prevent surgical site infections.
Custom-made intramedullary solutions for complex deformities, utilizing additive manufacturing to match patient-specific anatomy perfectly.

The PFNA Proximal Femur Interlocking Intramedullary Nail is engineered for the highest stability in trochanteric fractures. Its unique helical blade design provides excellent compaction of the cancellous bone, ensuring superior grip and resistance against cut-out, especially in elderly patients with poor bone quality.
When international medical distributors and hospital groups search for an Intramedullary Nails Exporter, their procurement criteria are rigorous. In the modern medical landscape, "Price" is no longer the only factor. Global buyers are prioritizing:
Compliance with CE (MDR) and ISO 13485 is non-negotiable. Implants must undergo strict biocompatibility testing and mechanical fatigue testing to ensure safety over years of implantation.
Demand is surging for Type II Anodized Titanium nails. As a factory, we use premium raw materials that offer the best balance of flexibility (to prevent stress shielding) and strength.
An IM nail is only as good as the instruments used to implant it. Procurement managers look for "All-in-one" solutions where the nail system is accompanied by intuitive, ergonomic surgical sets.
Post-pandemic, the ability to maintain consistent stock and provide rapid lead times has become a competitive advantage for Chinese manufacturers over European counterparts.
Changsha WEX Medical Instruments Co., Ltd. represents the peak of Chinese medical manufacturing. We combine cost-efficiency with uncompromising precision engineering.
We utilize advanced CNC sliding headstock auto-lathes and 5-axis machining centers to ensure every nail's thread and locking hole is accurate to the micron.
Our packaging and final inspection are conducted in Class 100,000 cleanrooms, adhering strictly to GMP standards to ensure zero contamination.
We partner with global brands to provide custom-label manufacturing, allowing our clients to leverage our R&D and production capacity.
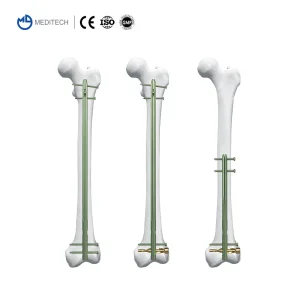
The Titanium Orthopedic Surgical Implant Suprapatellar Approach Tibial Intramedullary Nail is designed for versatile surgical approaches. Whether using the infrapatellar or the increasingly popular suprapatellar approach, our nail provides unparalleled ease of insertion and multi-planar locking options for distal fractures.
In high-volume trauma hospitals, our interlocking nails provide the reliability needed for emergency fracture fixations, ensuring rapid surgical turnover and consistent results.
For elderly patients with hip fractures, our PFNA and Gamma nail systems provide the specialized fixations required for osteoporotic bone, reducing the risk of secondary surgeries.
Athletes requiring rapid return to activity benefit from our humerus and tibial nails, which allow for early weight-bearing and functional rehabilitation.
Changsha WEX Medical Instruments Co., Ltd. has experiences of more than two decades in areas of innovation and production of orthopaedic implants, such as bone plates, bone screws, interlocking nails, spine implants/internal fixation systems, and all kinds of instruments, which are branded by "WEX". All the implant products have certification of CE and ISO9001/ISO13485, so can reach different requirements of customers.
With a Quality Management System passed ISO 9001:2008 and ISO 13485:2003, we have bright and spacious factory workshops; more than hundred production equipments, such as advanced CNC machining centre machines, CNC sliding headstock auto-lathes, CNC milling machines, CNC lathing machines, upmarket ultrasonic cleaning equipments and etc; more than ten advanced and well-appointed testing equipment, such as Projectors, Tensile-testing machines, Durometers, Carbon and sulphur analyzers, Pitting LP detecting instruments and Fluorescent fault detectors and so on. The packaging process is conducted in clean workshop line with GMP standard.
At the beginning of company's establishment, we aim to design and produce implantable products professionally for our customers. Relying on the operational systems of development, design, production and marketing, which is formed by the orthopaedic experts in domestic large and medium-sized hospitals, also we completed the clinical insurance for our products.
We satisfy our customer's requirements with best quality and best service, based on our advanced production and processing technology. Because we insist on the idea of "Be professional for manufacturing high-quality products, be wholehearted for serving customers."

