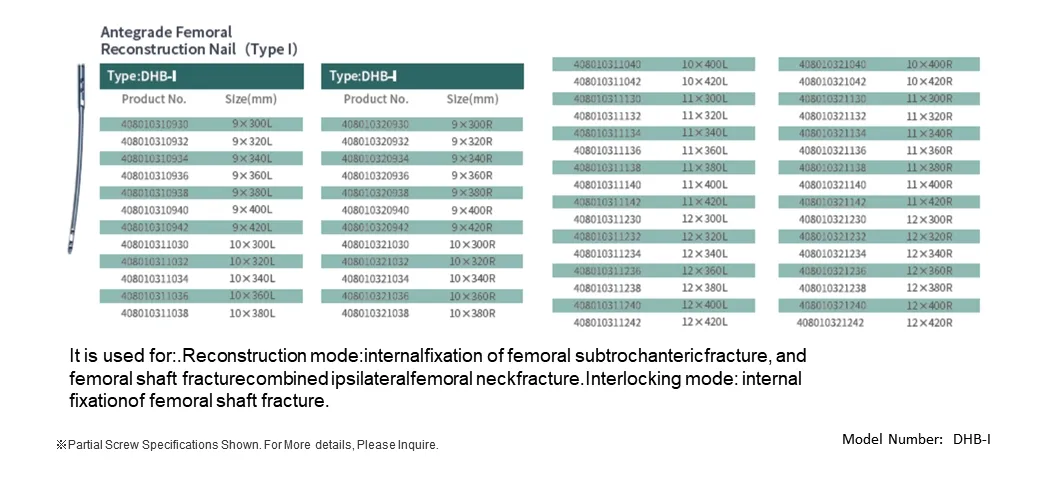
Product Introduction
The intramedullary nail system is designed to be implanted within the medullary canal, forming a composite structure with the bone to share mechanical loads. Positioned along the central axis of the femoral shaft, the nail offers optimal resistance to bending forces. Proximal and distal locking screws ensure stable fixation, allowing axial compression while preventing rotational instability. This femoral reconstruction nail system, composed of the main nail, traction screw, locking screws, and end cap, is made entirely from TC4 titanium alloy. It is primarily intended for femoral shaft fractures and complex femoral injury patterns.
Key Features
- Two configurations available: Reconstruction mode and Interlocking mode
- 5° lateral offset and 1500 mm anterior bow for anatomical alignment
- Flat lateral surface with low-profile groove design to minimize soft tissue irritation
- Multi-angle, multiplanar distal locking for enhanced anti-rotational stability
Core Advantages
- Minimizes soft tissue irritation to the lateral vastus muscle via proximal cutting design
- Dual surgical mode selection to adapt to fracture type and complexity
- Optimized geometry enhances anatomical fit and mechanical strength
- Advanced locking design improves fixation in complex or unstable fractures
Clinical Application
Indicated for femoral shaft fractures, femoral neck fractures, and complex combined fractures such as:
- Ipsilateral femoral shaft and neck fractures (AO/OTA 32-A/B/C with 31-B)
- Subtrochanteric fractures (31-A1B3)
- Intertrochanteric fractures combined with femoral shaft fractures
Professional Manufacturing
We are a leading manufacturer of standardized orthopedic implants and instruments. Our product range covers trauma, spine, joints, sports medicine, and CMF. With in-house R&D, certified laboratories, and modern production lines, we focus on reliable, scalable manufacturing to support global distributors and importers.
Frequently Asked Questions
What shipping methods do you offer and what are the costs?
We typically utilize courier services for shipping, but can accommodate postal services upon request. Costs are determined by destination, package size, and weight; a detailed quote is provided once specific requirements are known.
Are your orthopedic products certified?
Yes, our manufacturing facility holds the EN ISO 13485 certification, ensuring all medical devices meet stringent requirements for safety, quality, and regulatory compliance.
What is the Minimum Order Quantity (MOQ)?
Our standard minimum order value is US$1000. We also offer samples at cost for potential clients to evaluate product quality before committing to larger orders.
Can you manufacture custom products not listed in the catalogue?
Yes, we provide OEM/ODM services. If you provide pictures or physical samples, we can work with you to create custom solutions, though lead times may be longer.
Do you maintain stock for immediate shipment?
We maintain stock for most catalogue items with regular monthly production cycles. However, stock levels vary based on demand, so please consult our team for real-time inventory updates.
What material is used for the Intramedullary Nail?
The entire system, including the main nail and screws, is manufactured from high-grade Titanium Alloy TC4 for optimal biocompatibility and mechanical strength.


















 WEX Medical
WEX Medical