



1 / 5










| Customization: | Available |
|---|---|
| Certification: | CE, ISO9001 |
| Disinfection: | Without Disinfection |


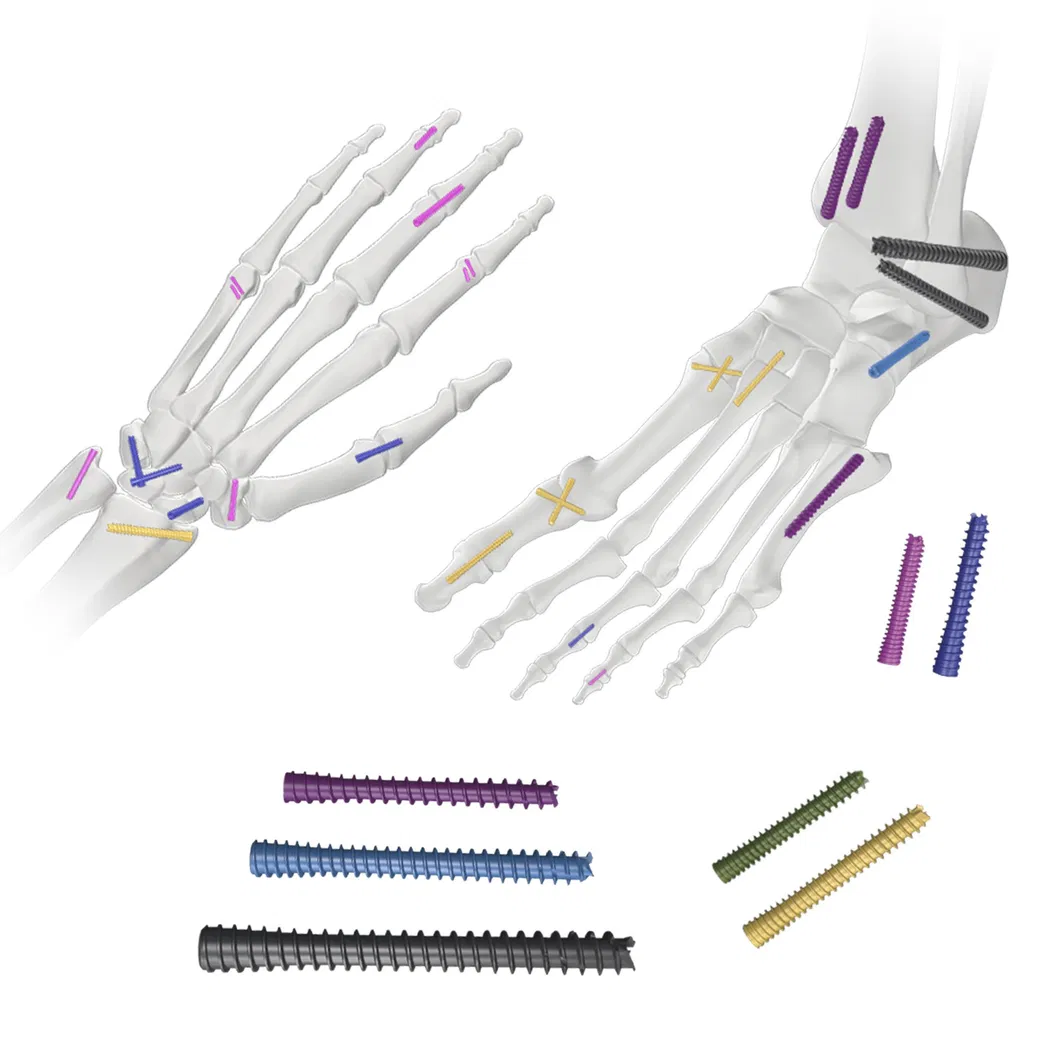

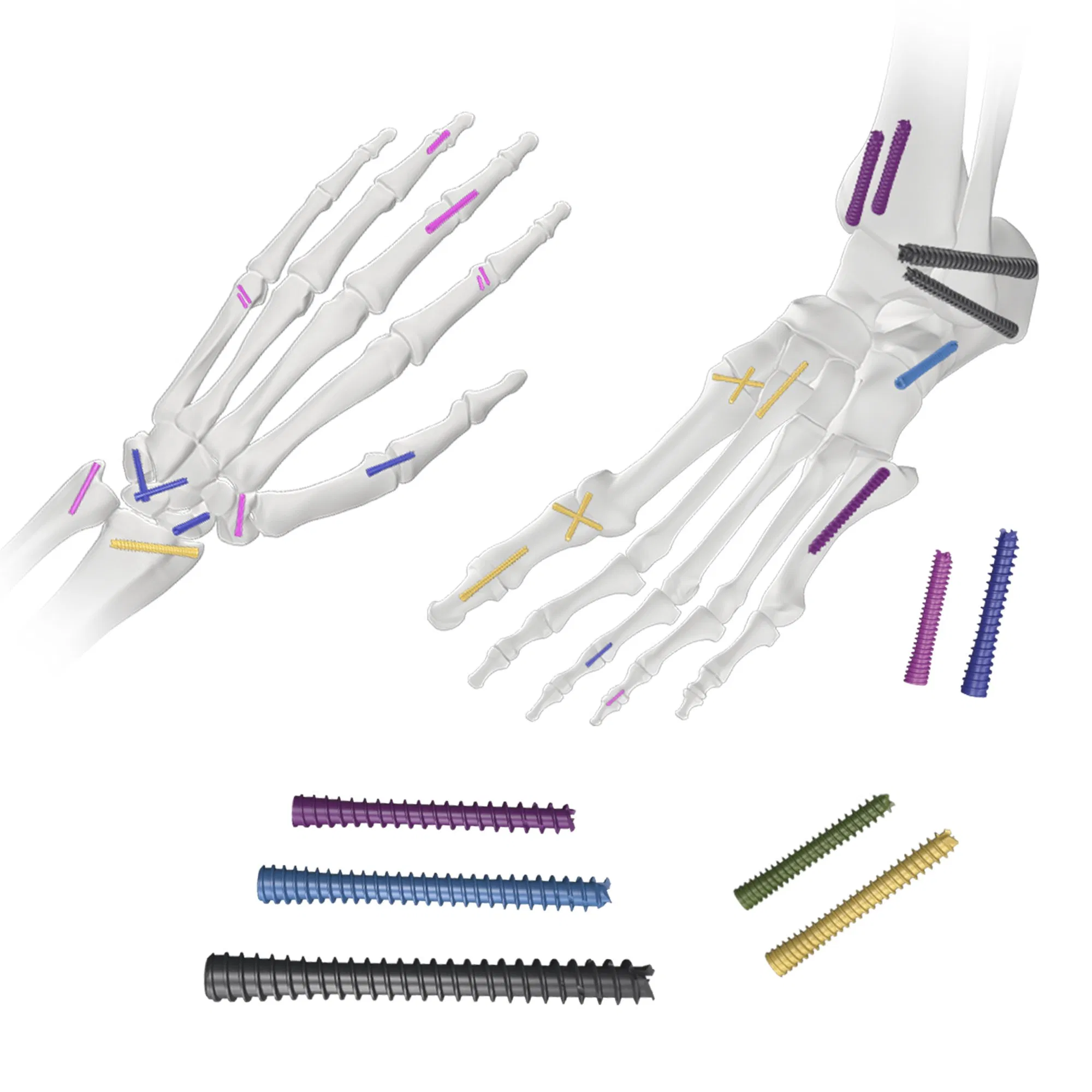
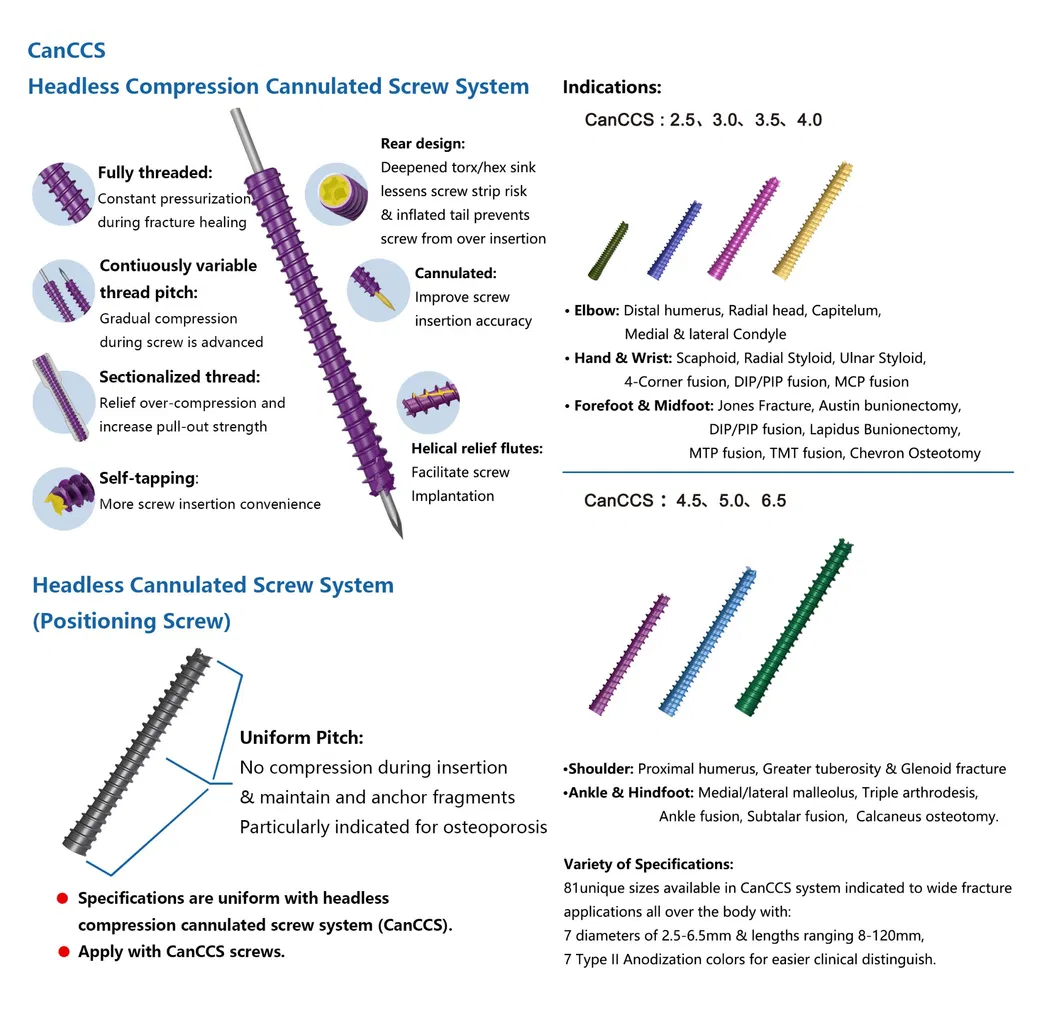
| Description | SPEC1 (Diameter) | SPEC2 (Length) |
|---|---|---|
| Headless Cannulated Screw | Ф2.5-Ф6.5 | 8-120 |





A leading medical device company founded in 1994 is now one of the largest designers, manufacturers, and suppliers of orthopedic implants and instruments. With over 60,000 square meters of workshop, the facility serves as a long-term instrument contract manufacturer for top global orthopedic companies. Product lines include traumatology, spinal fixation systems, and surgical instruments, such as femoral nails, titanium plates, pedicle screws, and PEEK cages.

 WEX Medical
WEX Medical