


1 / 5










| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |
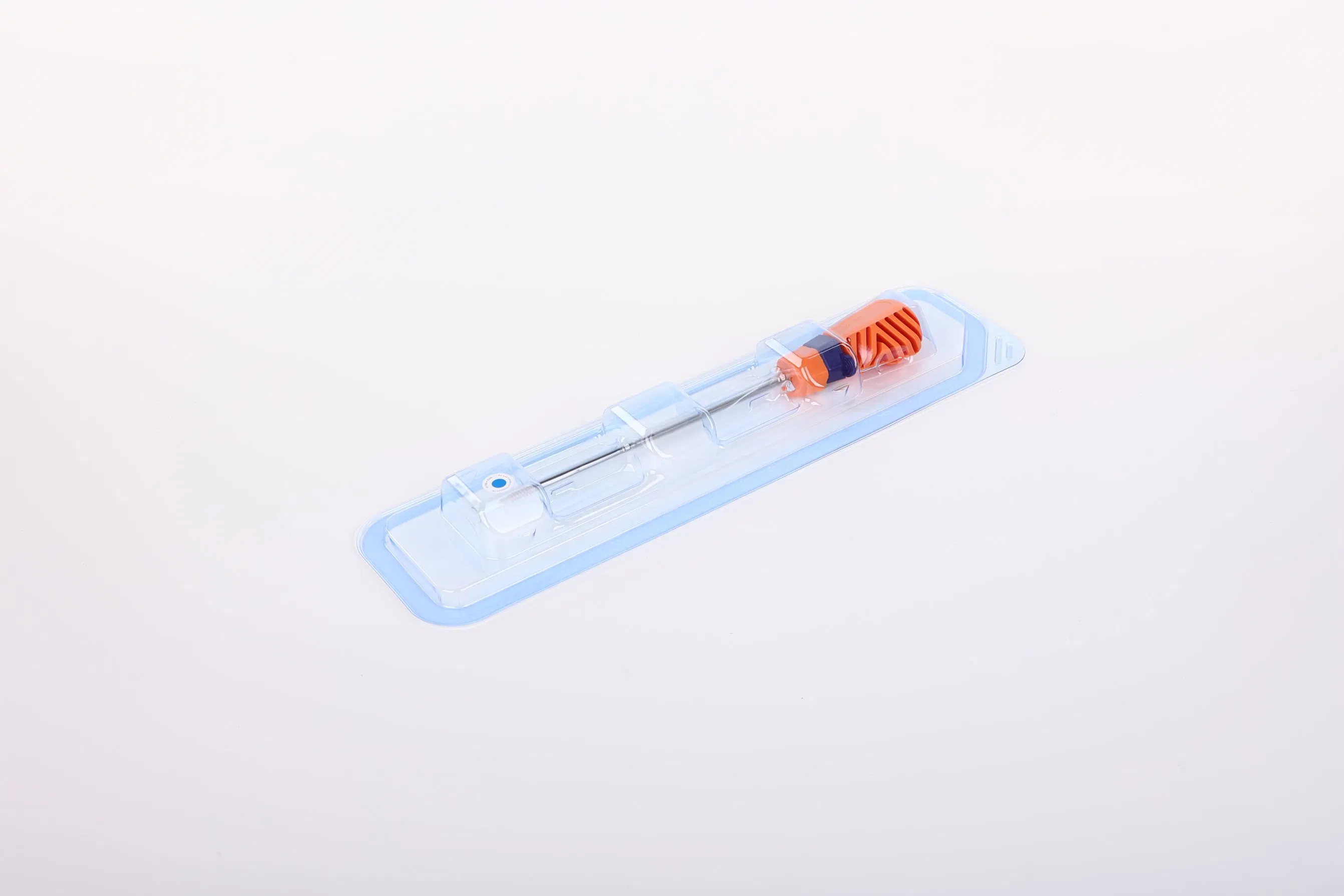
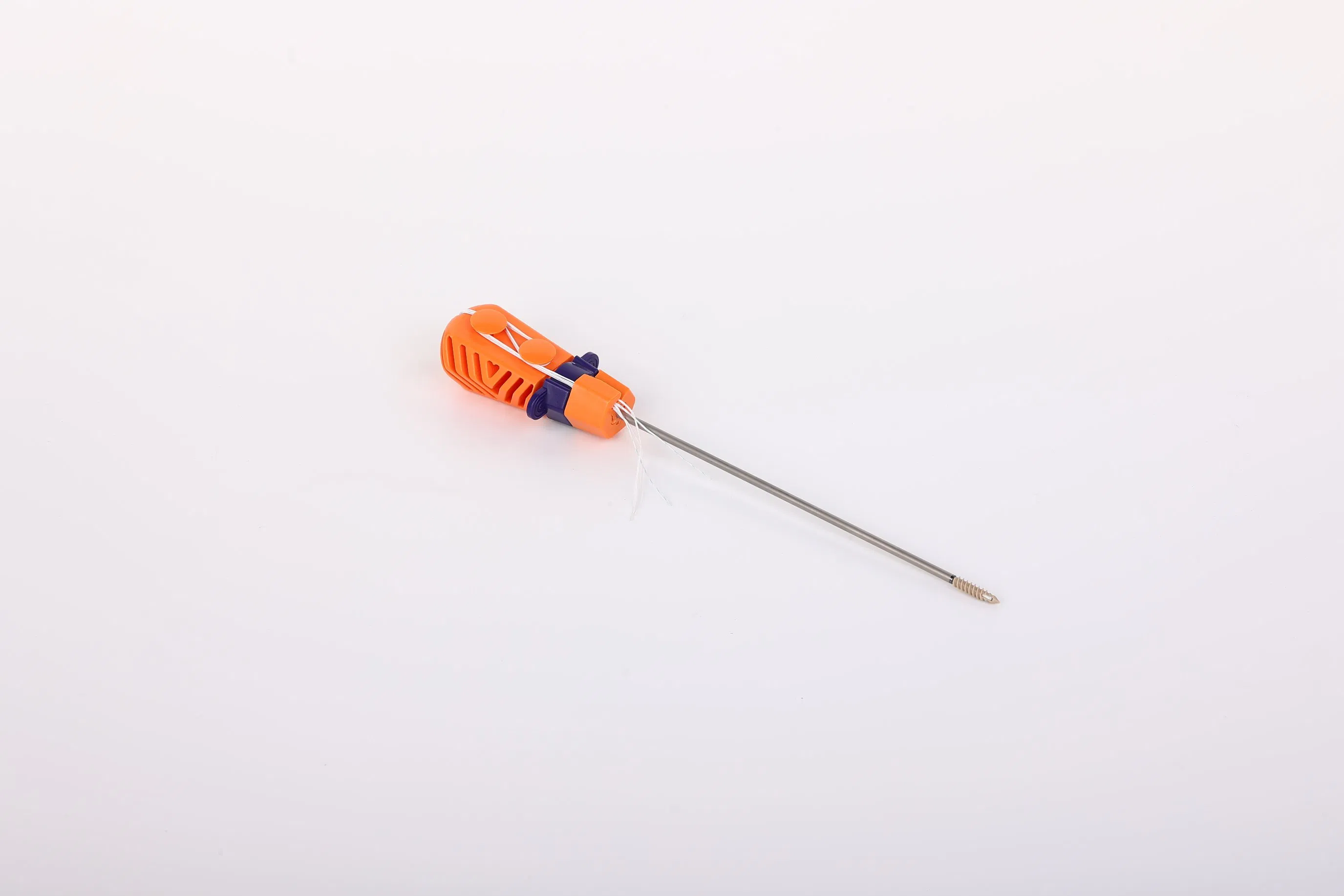
| Model NO. | WZ-MDT-D-05 |
| Certification | CE, ISO13485 |
| Color | Silver and Orange |
| Usage | Orthopedic Surgery |
| Sterile | Eo Sterile |
| Warranty | 3 Years |
| Function | Connect Soft Tissue and Bone |
| Material | Peek, Titanium Screw |
| Size Range | 1.8mm to 6.5mm |
| Origin | China |

System Components: The Suture Anchor System consists of an anchor, non-absorbable suture, and inserter.





 WEX Medical
WEX Medical