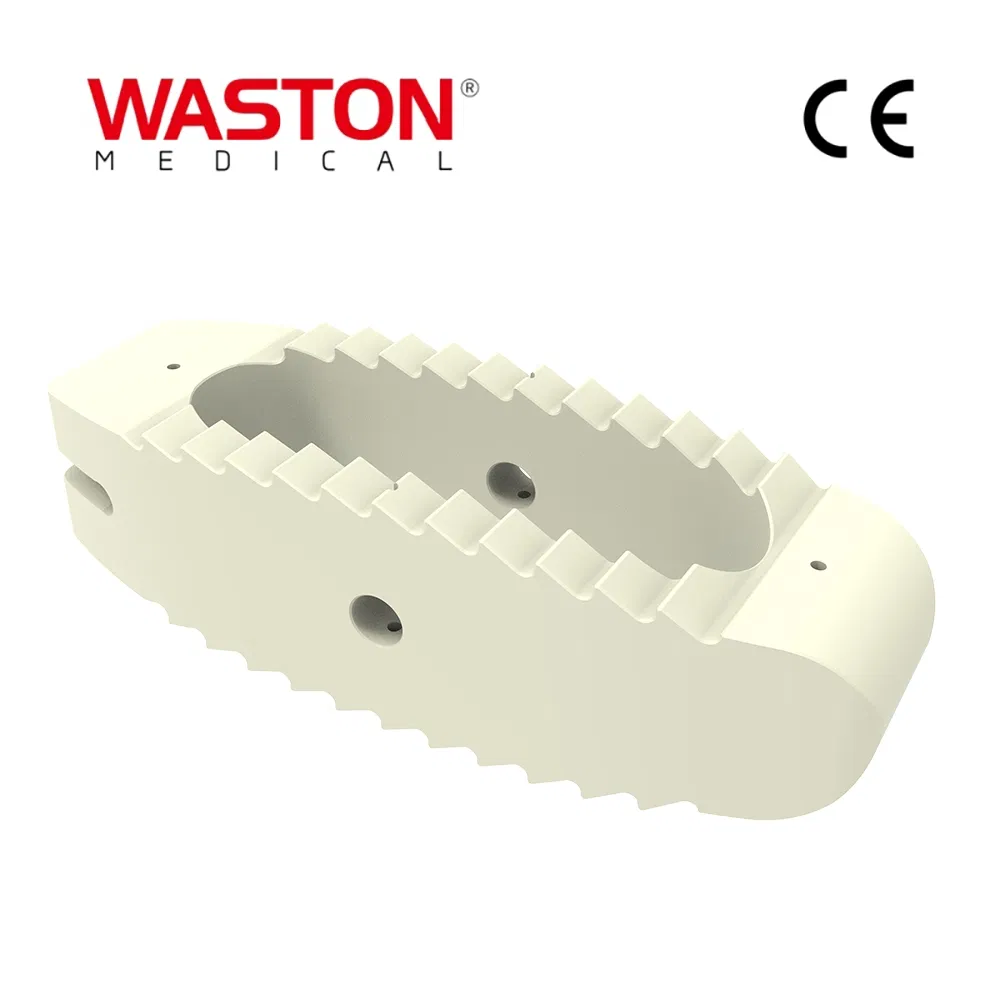
As a national high-tech enterprise, R&D investment consistently exceeds 8% of total sales. We transform clinical experience into innovative achievements, pioneering revolutionary technologies in fixation systems, rib plates, and spinal implants endorsed by global markets.
Operating strictly under the ISO13485 quality system and CE MDD 93/42/EEC directives, we utilize advanced machining centers imported from Germany, USA, and Japan. Our testing center includes biomechanical and physical-chemical laboratories to ensure strict raw material control and product performance analysis.
Global Presence & Certifications
Our products are exported to numerous countries including France, Turkey, Indonesia, Mexico, Russia, and Thailand. We also serve as a major OEM supplier to leading global players in the medical device industry.
Frequently Asked Questions
Is customized design available for orthopaedic implants?
Yes, we provide customized design solutions based on specific requirements and clinical needs.
What is your quality certification status?
We hold CE and ISO certifications, and FDA approval for our Spinal System products.
Can I request samples for quality and market testing?
Yes, we welcome sample orders to test our product quality and service before full-scale procurement.
What are the typical lead times for orders?
Sample orders are typically processed within 7 days, while standard bulk orders are delivered within 25 days.
Do you maintain inventory for regular orthopaedic items?
Generally, we maintain stock for listed products. However, availability depends on current sales conditions; please consult our sales team for real-time inventory information.
How do you handle defective goods?
For any verified defective goods, please provide photographic evidence for confirmation, and we will arrange for replacements.















 WEX Medical
WEX Medical