Basic Information
Model NO.
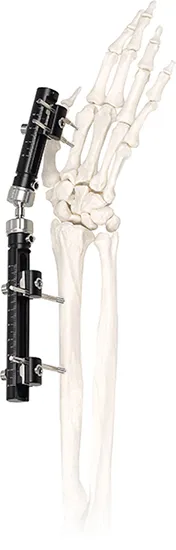
External Fixation
Feature
Reusable
Certification
CE, ISO13485
Color
White & Black
Usage
Bone Fixation
Material Type
Titanium Alloy & Stainless Steel
Name
External Fixator
Origin
China
HS Code
90211000
Production Capacity
500,000 Pieces/Year
Frequently Asked Questions
Q1: What are the primary materials used in these external fixators?
A1: Our external fixators are manufactured using high-grade Titanium Alloy and Stainless Steel to ensure biocompatibility and structural integrity.
Q2: Are the external fixation systems reusable?
A2: Yes, the feature of this model is that it is reusable, following standard sterilization protocols.
Q3: Which certifications do these medical devices hold?
A3: The products are fully certified with CE and ISO13485 standards, meeting international quality requirements for medical instruments.
Q4: Does the unilateral design offer advantages during surgery?
A4: Yes, the unilateral design is lightweight and stable, making it particularly effective for emergency cases and minimizing surgery time.
Q5: How does the "Minimal-Invasive" feature benefit the patient?
A5: Minimal-invasive surgery ensures there is no negative effect on the blood supply to the fracture area, which promotes faster healing and bone union.
Q6: Is a second surgery required to remove the fixator?
A6: No, one of the key benefits is that the fixator can be safely removed at a clinic without the need for a secondary surgical procedure.























 WEX Medical
WEX Medical