














| Still deciding? Get samples of $ ! US$ 108/Piece Request Sample |
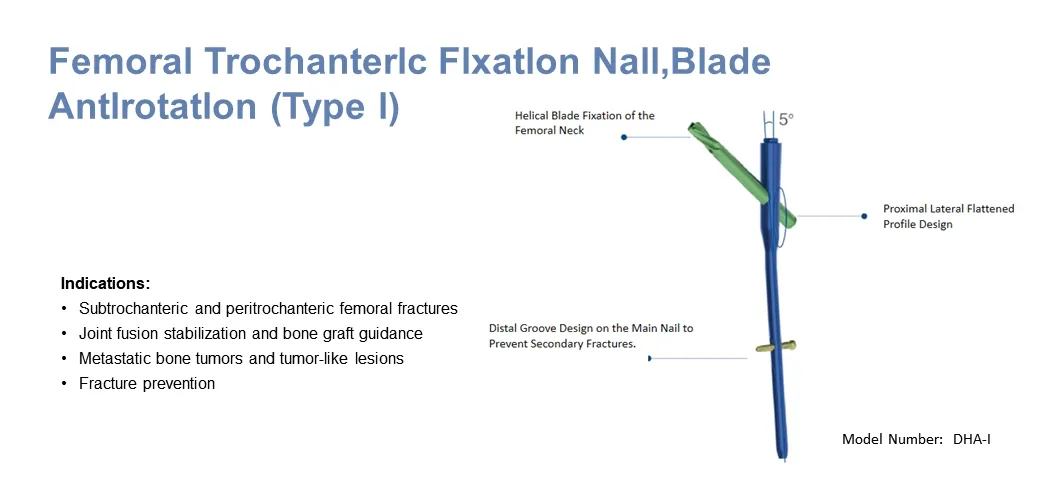
The PFNA (Proximal Femoral Nail Anti-rotation) is an advanced intramedullary implant specifically designed for the treatment of hip fractures, particularly unstable intertrochanteric and proximal femoral fractures.
This system achieves stable fixation by sharing the load with the bone through the intramedullary nail, significantly reducing the risk of implant loosening and failure. Made from titanium alloy, PFNA combines excellent biocompatibility with mechanical strength to ensure long-term durability and patient safety.


Professional Orthopedic Manufacturer
We are a leading manufacturer of standardized orthopedic implants and instruments for global distributors. Our full product range covers trauma, spine, joints, sports medicine, and CMF.
With in-house R&D, a certified lab, and modern production lines, we focus on reliable, scalable manufacturing to support high-volume distribution and clinical excellence.
 WEX Medical
WEX Medical