📋 Basic Info.
Usage
Correct Deformities, Fracture Fixation
Properties
Implant Materials & Artificial Organs
Application
Trauma Operation
Instrument Classification
Class II
After-Sale Service
Online Technical Support
Package
Sterile/Non-Sterile Package
Production Capacity
10000pices/Year
👥 Product Description
The Ilizarov External Fixator with Foot Ring is a specialized orthopedic device engineered based on the Ilizarov principle, designed for the treatment of complex foot and ankle conditions, including congenital deformities, traumatic fractures, nonunions, malunions, and post-traumatic contractures. This modular system integrates high-strength external fixation components with a customized foot ring, delivering unparalleled stability, adjustability, and minimally invasive treatment capabilities for orthopedic surgeons worldwide.

Crafted from medical-grade stainless steel and titanium alloy, the fixator features a durable, corrosion-resistant structure that ensures long-term reliability during extended treatment periods. The foot ring is anatomically contoured to fit the natural curvature of the foot, providing optimal contact and support without compromising soft tissue integrity. Its modular design allows for flexible assembly, enabling surgeons to tailor the fixation configuration to each patient's unique anatomical needs and treatment goals—whether for gradual deformity correction, fracture stabilization, or limb length discrepancy management.
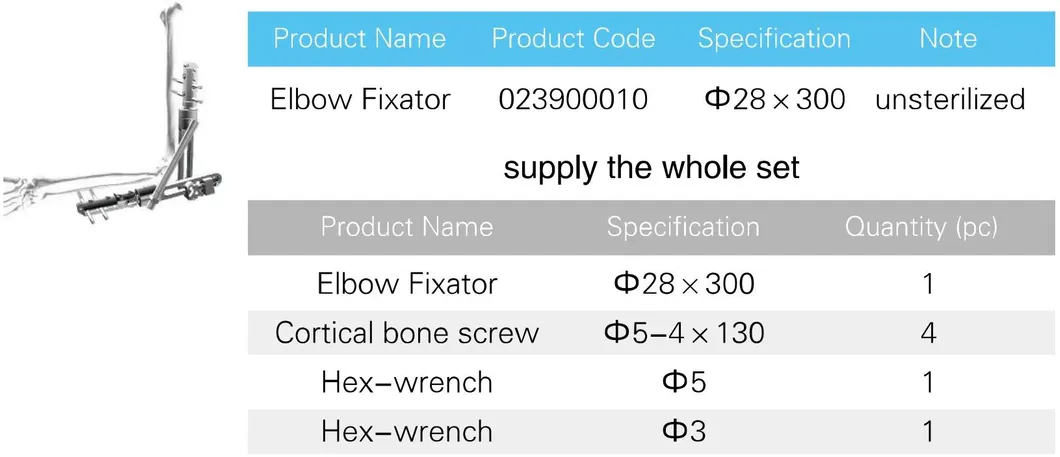
*All the prices are only for reference. Exact price will be based on the latest quotation.
| Product Name |
Specification |
Size |
Quantity |
| Dissection IV(Φ8) Femur External Fixation |
Carbon Fiber Rod |
Φ8×250 |
2 |
| Rod-rod Coupling |
- |
4 |
| Pin-post Coupling |
- |
2 |
| Straight Post/30°Angled Post |
- |
4 |
| One Step Pin |
Φ5×180 |
4 |

❓ FAQ
What are the primary clinical applications of this fixator?
It is primarily used for correcting foot and ankle deformities, traumatic fractures, nonunions, and limb length discrepancies using the Ilizarov principle.
What materials are used in the construction of the device?
The system is crafted from high-quality medical-grade stainless steel and titanium alloy to ensure durability and corrosion resistance.
Does the product come with any certification?
Yes, the product is CE and ISO13485 certified, meeting international standards for Class II medical instruments.
Is the fixator provided in sterile packaging?
We offer both sterile and non-sterile packaging options depending on your specific surgical requirements.
What kind of after-sales support is available?
We provide one year of warranty and ongoing online technical support to assist with any operational or assembly queries.
Is OEM service available for this product?
Yes, OEM services are available to meet specific customization needs for different orthopedic requirements.






















 WEX Medical
WEX Medical