


1 / 5










| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |


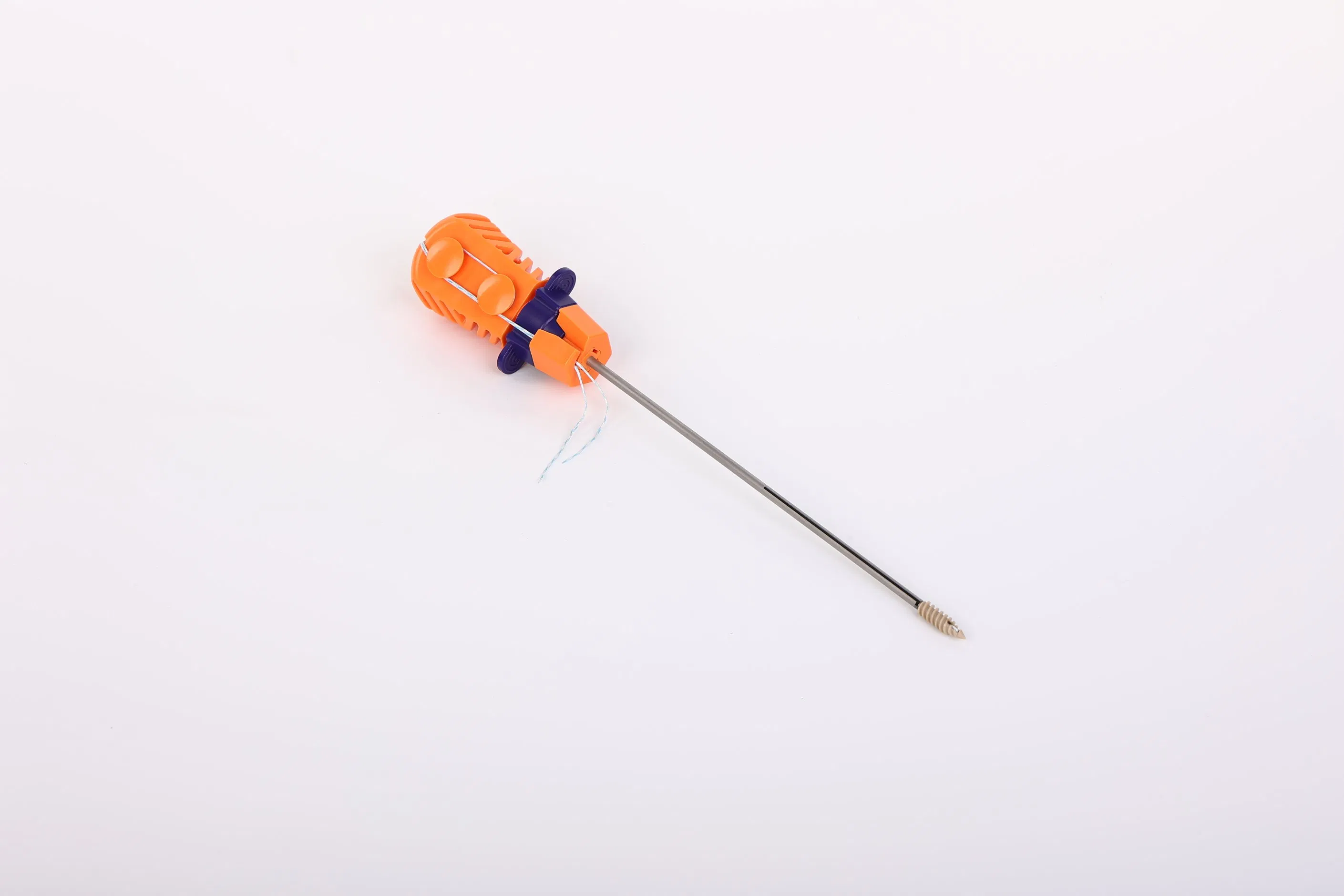
Suture Anchor System consists of an anchor, nonabsorbable suture, and an inserter. The system is designed for high-performance orthopedic applications:



Our product range includes Disposable Surgical Lavage Systems, Suture Anchor Systems, Meniscal Repair Systems, and other orthopedic surgical consumables. To guarantee quality and safety, we utilize:
 WEX Medical
WEX Medical