












| Still deciding? Get samples of $ ! US$ 80/Piece Request Sample |
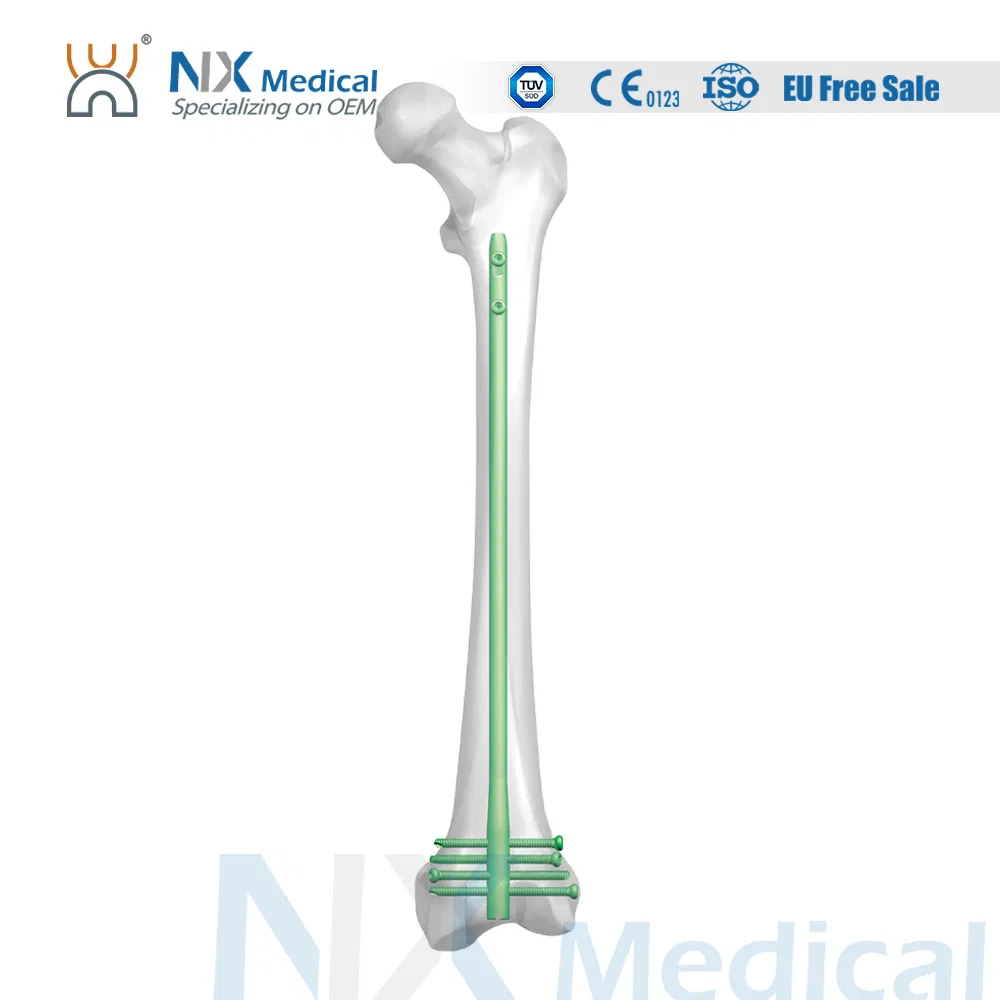
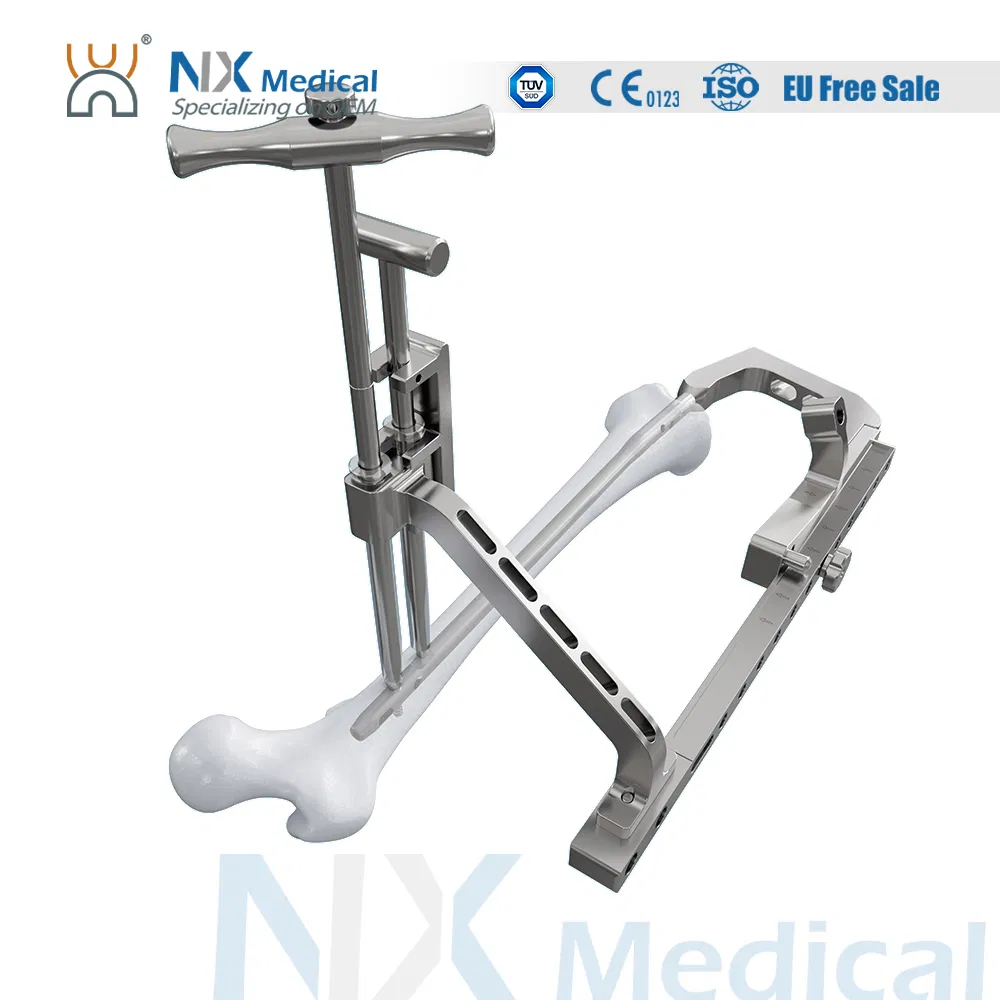
The Retrograde Femoral Nail System is designed for stable internal fixation of femoral fractures, specifically focusing on the distal femoral area. This system integrates advanced engineering to ensure surgical precision and patient recovery.


| Product Name | Size / Length (mm) | Material |
|---|---|---|
| Retrograde Femoral Nail | 9/10/11/12 x 200 - 360 (20mm increments) | Titanium Alloy (TA) |
| IM Nail Interlocking Screw (φ4.8mm) | 26 / 28 / 30 ... up to 84mm | Titanium Alloy (TA) |
| End Cap | 0 / 5 / 10 / 15 (Extension) | Titanium Alloy (TA) |
As a leading orthopedic product manufacturer in East China, we provide a comprehensive range of cost-effective surgical and reliable therapeutic solutions. Our facilities are committed to the global orthopedic industry through continuous investment in R&D and high-precision CNC machining.



Our quality systems fulfill strict self-audits and comply with international standards including ISO13485 and CE0123 certification. We ensure the safest and most advanced implants for rapid recovery and pain-free life after surgery.

 WEX Medical
WEX Medical