


1 / 5










| Still deciding? Get samples of $ ! US$ 10/Piece Request Sample |
| Model | Order No. | Item No. | Specification |
|---|---|---|---|
| Maxillofacial Micro X-Plate | 10.01.01.04021000 | 0539 | 4 holes, 0.6mm, 14mm |
| Maxillofacial Micro Rectangular Plate | 10.01.01.04023000 | 0672 | 4 holes, 0.6mm, 14*14mm |
| Maxillofacial Micro Straight bridge Plate | 10.01.01.04011020 | 0017 | 4 holes, 0.6mm, 20mm |
| Maxillofacial Micro Straight Plate | 10.01.01.04011015 | 0001 | 4 holes, 0.6mm, 16mm |
| 10.01.01.06011000 | 0002 | 6 holes, 0.6mm, 24mm | |
| 10.01.01.08011000 | 0003 | 8 holes, 0.6mm, 32mm | |
| 10.01.01.10011000 | 0004 | 10 holes, 0.6mm, 40mm | |
| 10.01.01.12011000 | 0005 | 12 holes, 0.6mm, 48mm | |
| 10.01.01.14011000 | 0671 | 14 holes, 0.6mm, 56mm | |
| Maxillofacial Micro Arc Plate | 10.01.01.06013000 | 0006 | 6 holes, 0.6mm, 23mm |
| 10.01.01.08013000 | 0007 | 8 holes, 0.6mm, 30mm | |
| 10.01.01.10013000 | 0008 | 10 holes, 0.6mm, 37mm | |
| 10.01.01.12013000 | 0009 | 12 holes, 0.6mm, 43mm | |
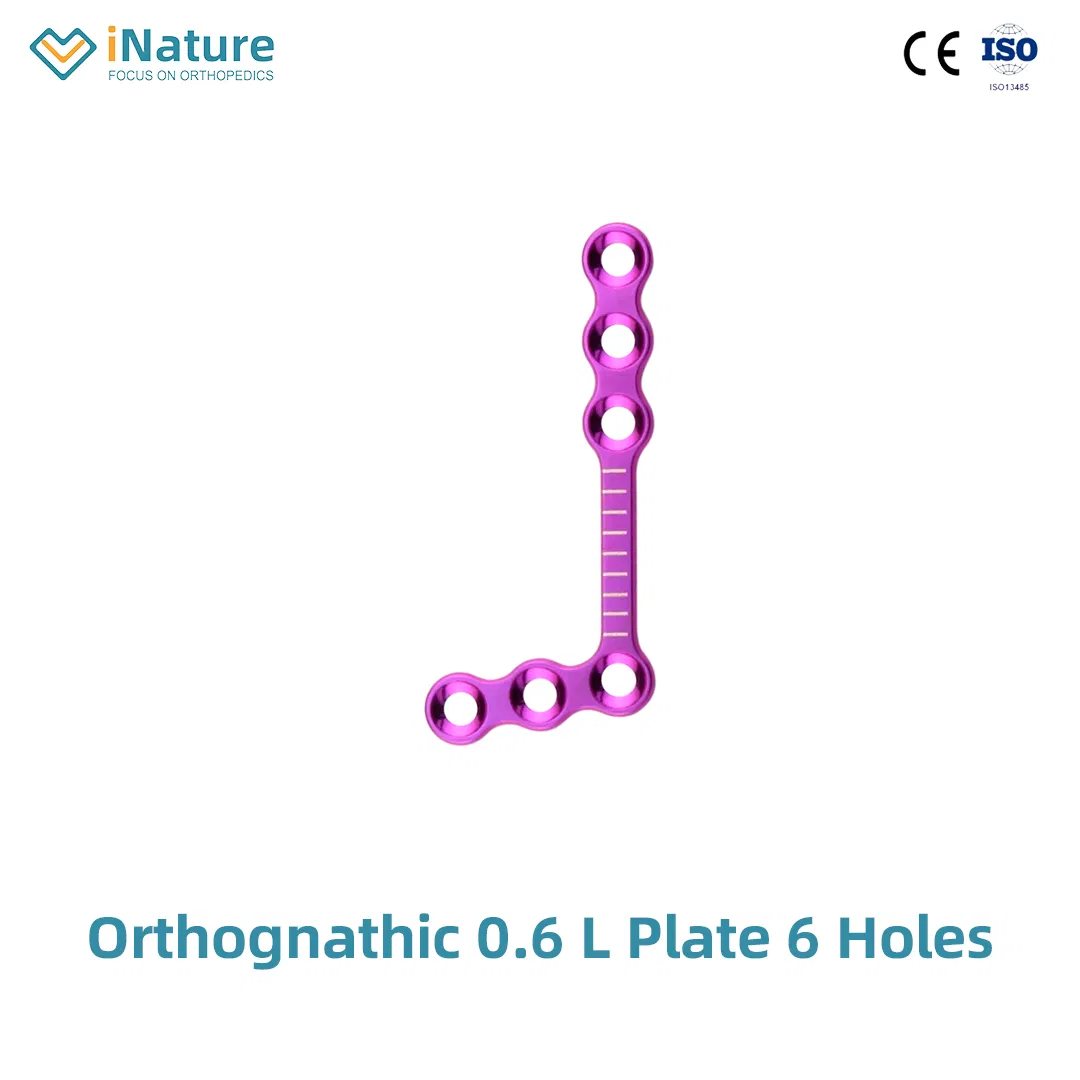
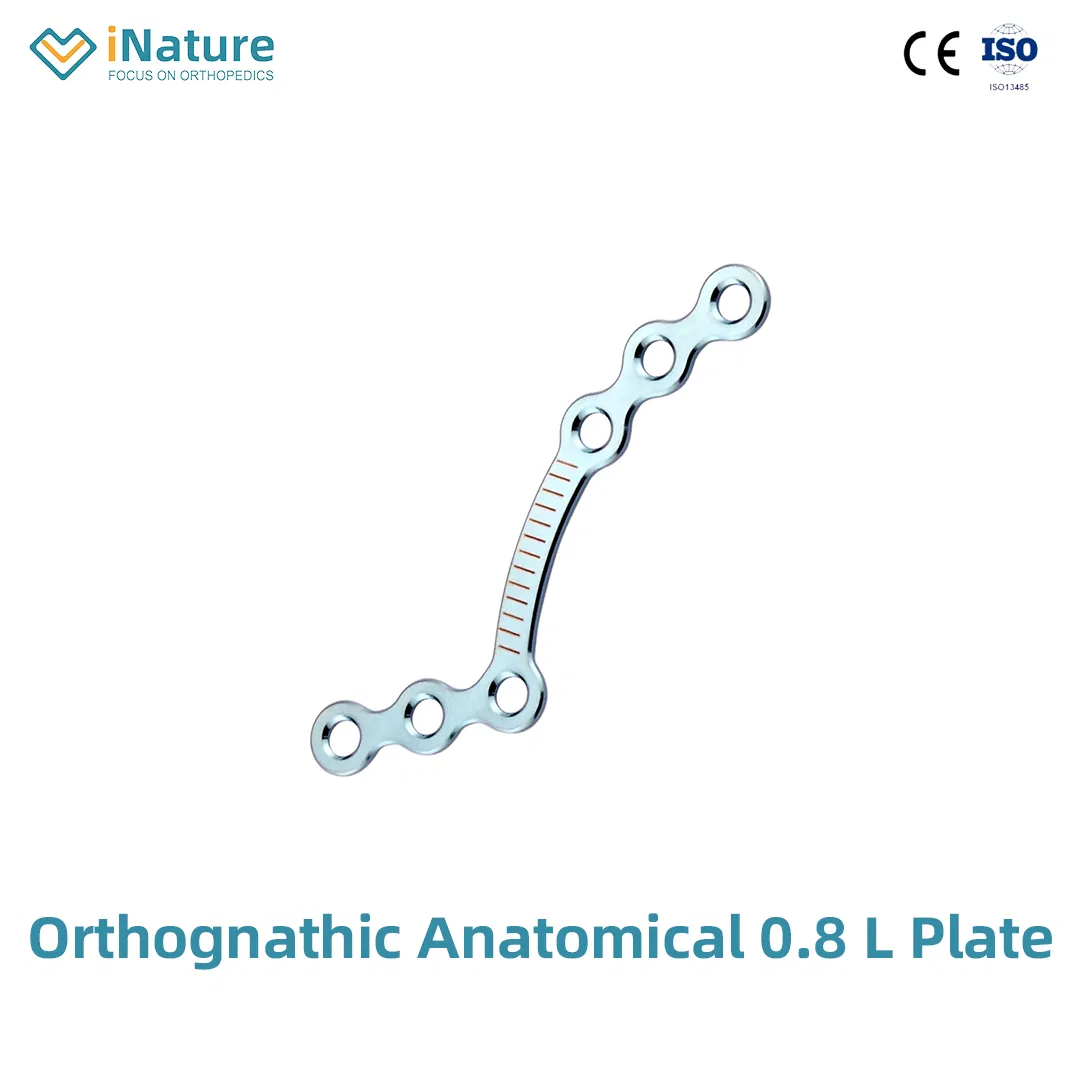
| Maxillofacial Micro 90°L-plate | 10.01.01.04115000 | 0010 | 4 holes, 0.6mm, 16mm, Left |
| 10.01.01.04215000 | 0011 | 4 holes, 0.6mm, 16mm, Right | |
| Maxillofacial Micro 110°L-plate | 10.01.01.04116000 | 0012 | 4 holes, 0.6mm, 16mm, Left |
| 10.01.01.04216000 | 0013 | 4 holes, 0.6mm, 16mm, Right | |
| 10.01.01.05116000 | 0535 | 5 holes, 0.6mm, 20mm, Left | |
| 10.01.01.05216000 | 0536 | 5 holes, 0.6mm, 20mm, Right | |
| Maxillofacial Micro Y-plate | 10.01.01.06021000 | 0538 | 6 holes, 0.6mm, 17mm |




 WEX Medical
WEX Medical