




1 / 5










| Still deciding? Get samples of $ ! US$ 24/Piece Request Sample |
| Product Number | Product Name | Material | Size(mm) |
|---|---|---|---|
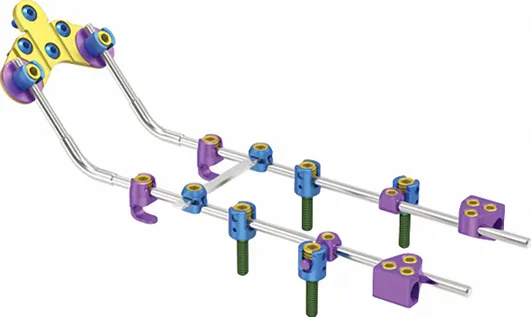
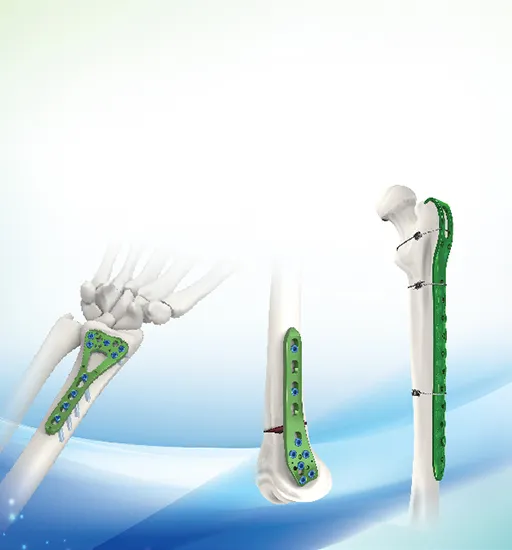
| 73601 | KCO Laminar Hook 736 | Titanium Alloy | 4.5mm |
| 73602 | 6mm |





 WEX Medical
WEX Medical