
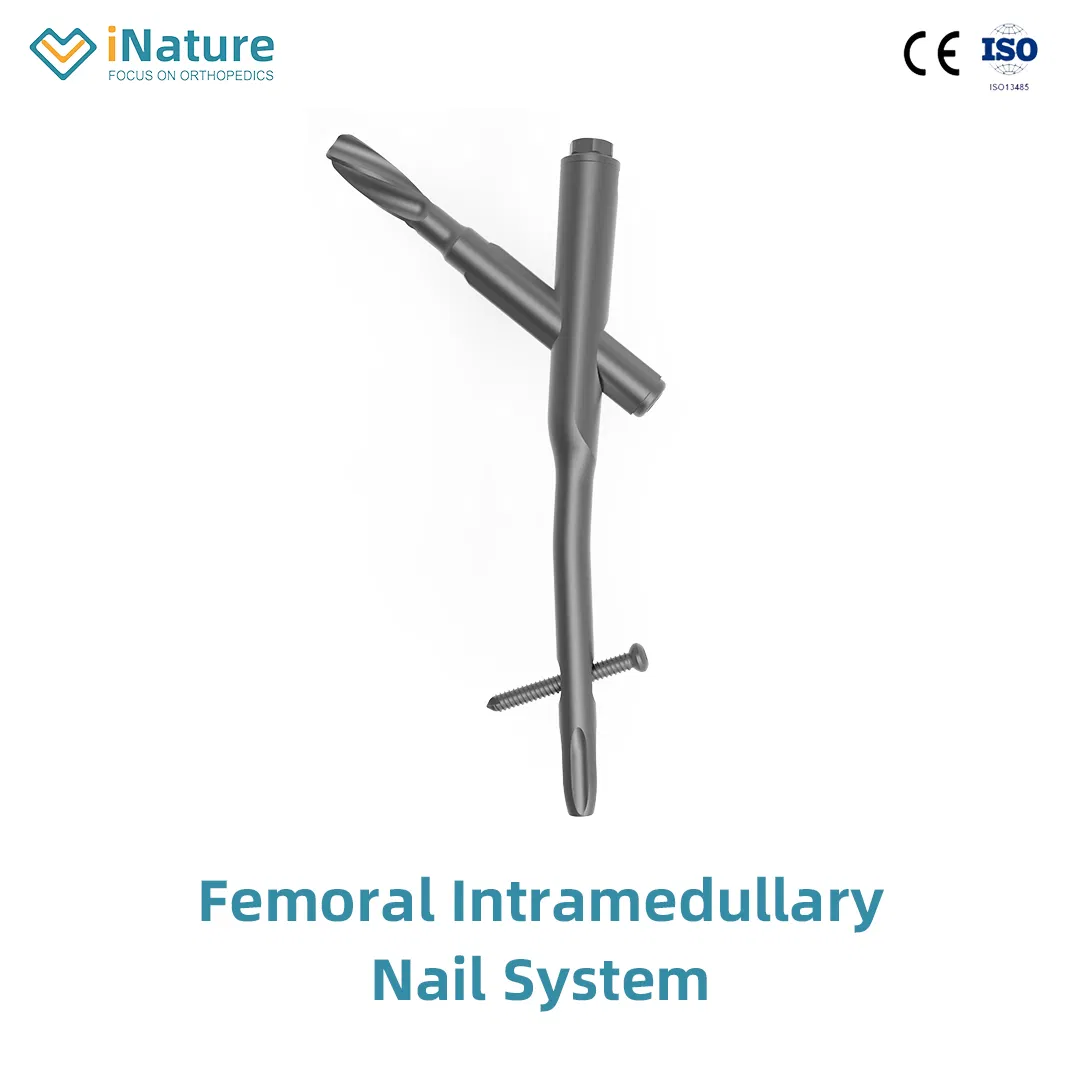



1 / 5










| Still deciding? Get samples of $ ! US$ 120/Piece Request Sample |
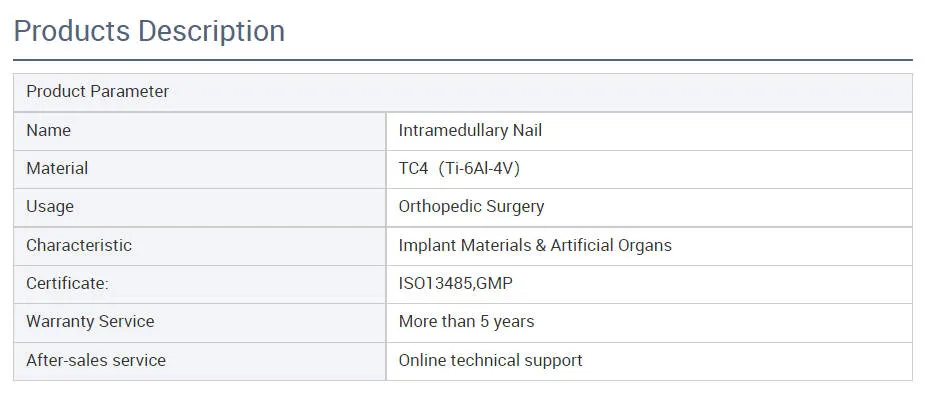
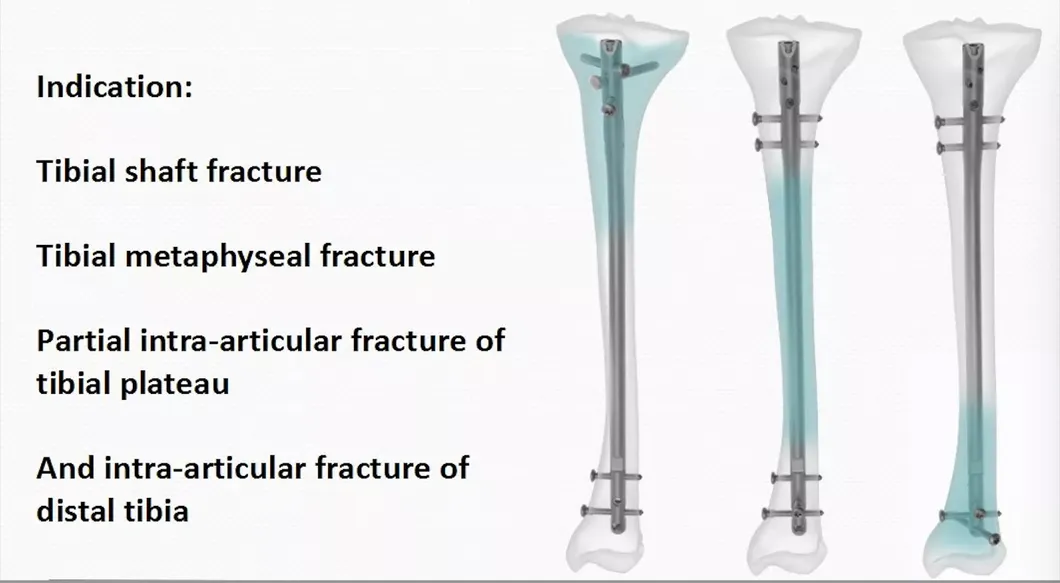

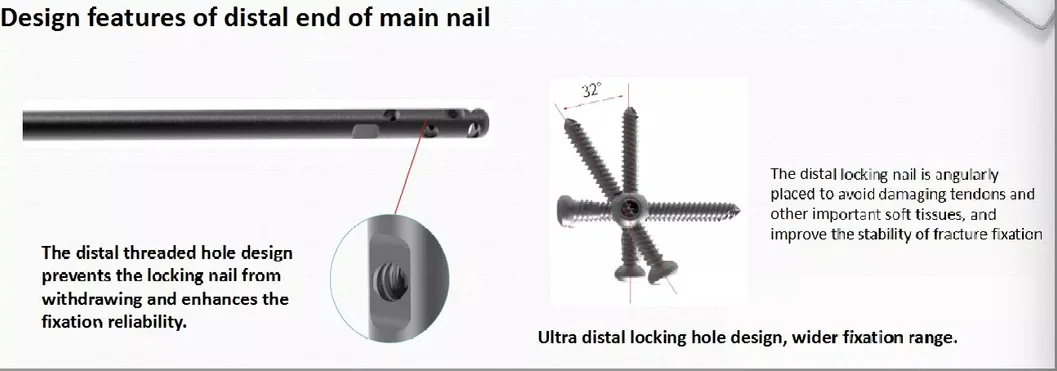

We provide three key guarantees starting from the date of sale:
| One Month | Return & exchange guarantee if product has quality problems (excluding operational errors). |
| One Year | Free repair service (shipping costs apply if caused by wrong operation). |
| After One Year | Timeless maintenance service with replacement parts and freight charged at cost. |
*Note: Prices are for reference. Exact prices are based on the latest quotation.




 WEX Medical
WEX Medical