Basic Information
Certification
CE, ISO13485
Capacity
50,000 pieces/Month
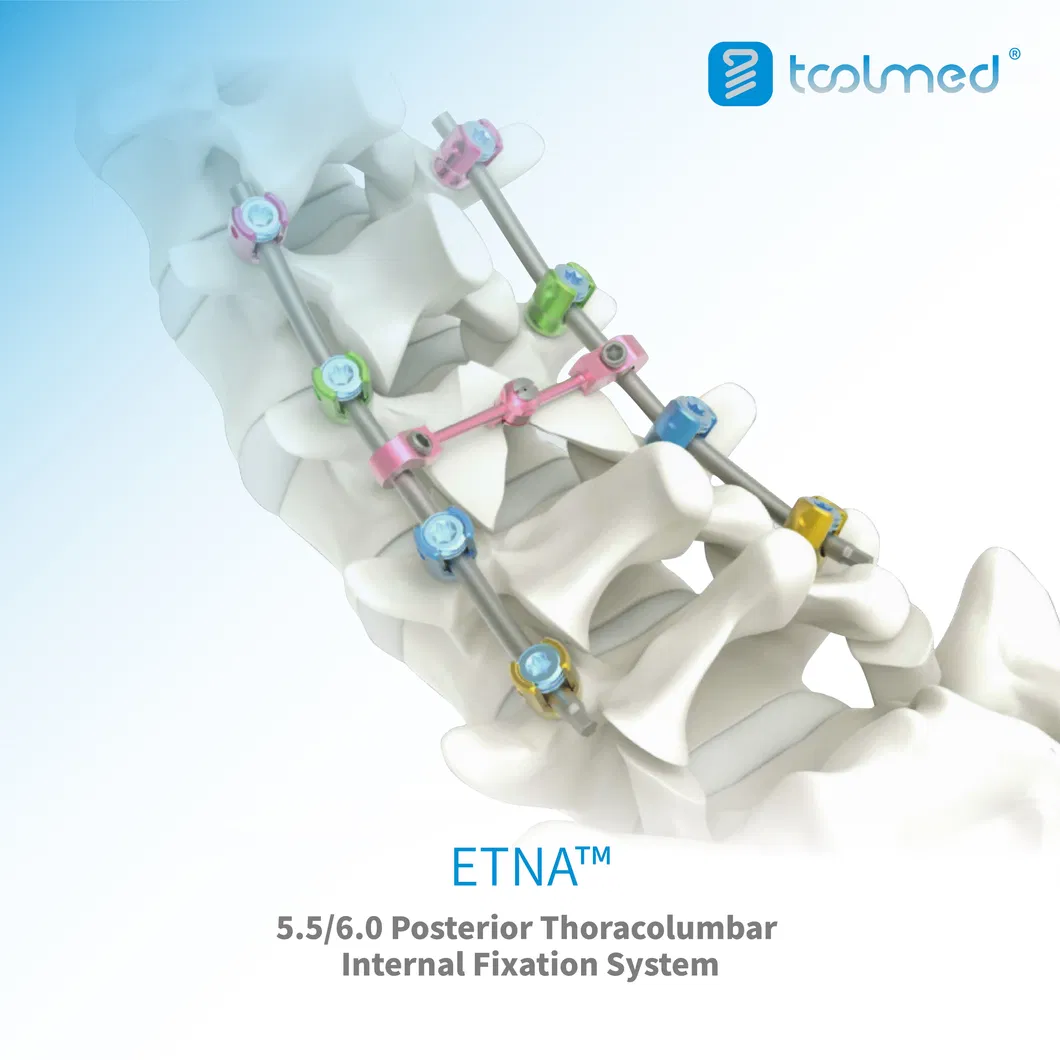
Key Advantages
-
Enhanced Stability and Secure Fixation
The integrated locking screws prevent cage migration and subsidence, promoting a stable environment for successful bone fusion.
-
Excellent Biomechanical Compatibility
The PEEK material's elasticity mimics natural bone, reducing stress shielding and the risk of adjacent segment disease.
-
Streamlined Surgical Procedure
The self-locking design simplifies implantation, potentially reducing operating time and improving surgical efficiency.
Clinical Applications
-
Primary Application in ACDF Surgery
Used to restore disc height and stabilize the spine following discectomy for treating cervical disc herniation or spondylosis.
-
Management of Cervical Instability and Stenosis
Provides structural support in cases of degenerative instability, stenosis, or spondylolisthesis to decompress neural elements.
-
Facilitation of Spinal Fusion
The cage creates a stable interbody space packed with bone graft to promote biological fusion between vertebrae.
Manufacturing & Quality Control
Frequently Asked Questions
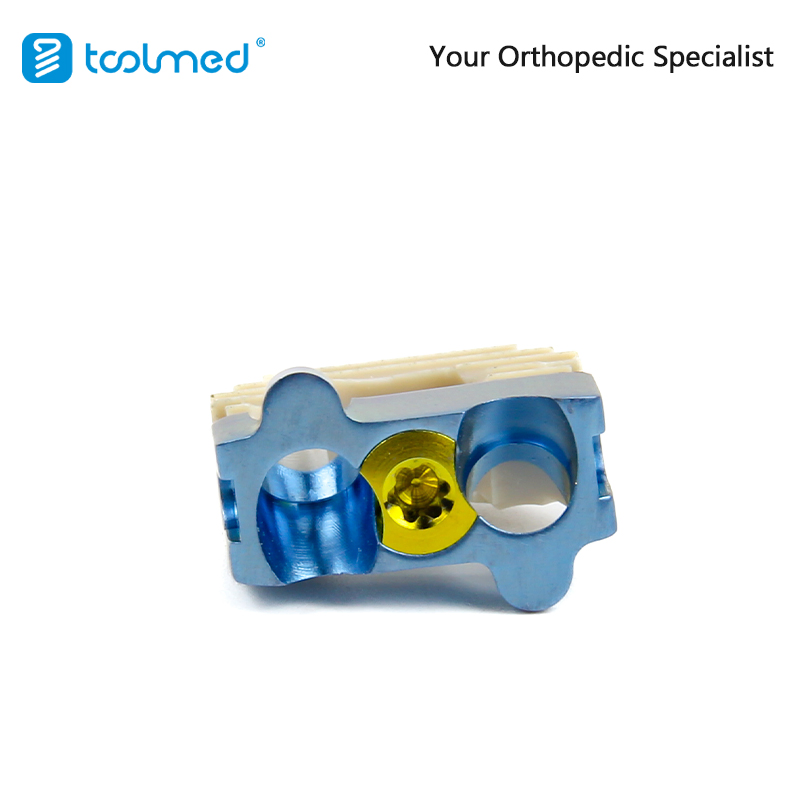
Q1: What materials are used in the cervical interbody fusion cage?
The cage is typically manufactured using high-quality PEEK (Polyetheretherketone) combined with Titanium Alloy locking screws for optimal biomechanical compatibility and stability.
Q2: Are these implants certified for international use?
Yes, the products are manufactured under strict quality systems and hold CE and ISO13485 certifications, ensuring they meet global medical standards.
Q3: What is the typical lead time for an order?
We maintain a consistent stock for standard specifications. Orders can generally be shipped within one week if stock is available.
Q4: Do you offer samples for clinical evaluation?
Yes, we provide samples for evaluation. Please contact our support team to discuss the specific requirements and freight arrangements.
Q5: What kind of warranty is provided for the orthopedic instruments?
We provide a comprehensive 5-year warranty for our products, reflecting our confidence in our manufacturing quality and durability.
Q6: Can the products be customized for specific surgical needs?
Yes, we provide OEM and ODM services to match special medical demands and specific regional requirements globally.

























 WEX Medical
WEX Medical