Product Description
◈
Reliable Suture Anchor System for Orthopedic Shoulder Procedures
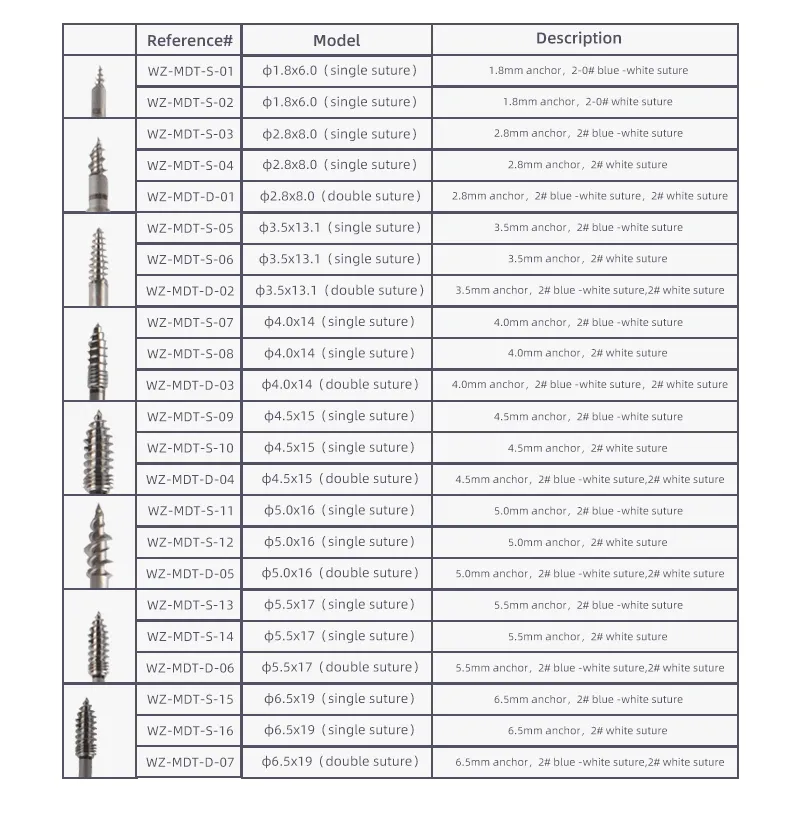
The Peek Suture Anchor System consists of an anchor, nonabsorbable suture, and inserter.
The Anchor is manufactured from high-grade Ti6Al4V alloy, adhering to ISO 5832-3:2016 standards. The suture is crafted from Ultra High Molecular Weight Polyethylene (UHMWPE) material without coating, meeting ASTM F2848-17 requirements. The blue suture is dyed using FDA-approved materials. The inserter component contacting the body is made of medical-grade stainless steel (ASTM F899-12b).
Frequently Asked Questions
Q1: What materials are used in the anchor?
The anchor is made of Ti6Al4V titanium alloy, which strictly follows the ISO 5832-3:2016 international standard for surgical implants.
Q2: Are the sutures medical grade?
Yes, the sutures are made of Ultra High Molecular Weight Polyethylene (UHMWPE) woven yarn, complying with ASTM F2848-17 standards, and the blue dye is FDA approved.
Q3: How is the product sterilized?
The suture anchor system is provided EO (Ethylene Oxide) Sterile, ensuring it is ready for immediate clinical use in orthopedic surgery.
Q4: What certifications does the product hold?
The product is certified under CE and ISO13485 standards, ensuring high quality and safety for medical applications.
Q5: What is the warranty period for this system?
The Suture Anchor System comes with a 3-year warranty period from the date of manufacture.
Q6: Is OEM service available?
Yes, OEM services are available to meet specific clinical or regional requirements.


















 WEX Medical
WEX Medical