●
Application: Widely used for different bone fracture of different bones.
●
Available Head: Hexagonal head.
●
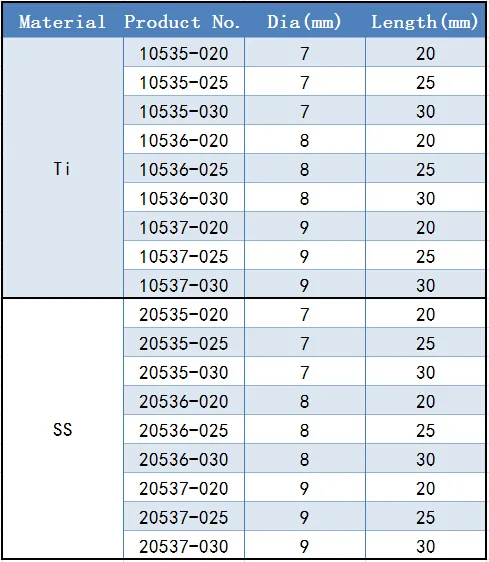
Diameter & Length:
7.0mm (20/25/30mm);
8.0mm (20/25/30mm);
9.0mm (20/25/30mm)
●
Instrument: ACL Interference Screw Instruments.
As a national high-tech enterprise, the proportion of R&D investment to total sales has been over 8% for consecutive years, realizing the transformation of experts' clinical experience and innovative ideas into achievements. Key revolutionary technologies include:
Rev Drill system, Three-Row-Stapler, Sternal Fixation and Rib plate.
Operating strictly according to the
ISO13485 quality system and
CE MDD 93/42/EEC directives.
Utilizing world-class equipment including German and American machining centers, and Japanese automatic lathes. The Testing Center includes:
- Biomechanical laboratory
- Physical-chemical testing center
Focused on three key points: raw material procurement control, product quality control, and new product performance analysis.

















 WEX Medical
WEX Medical