




1 / 5










| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |
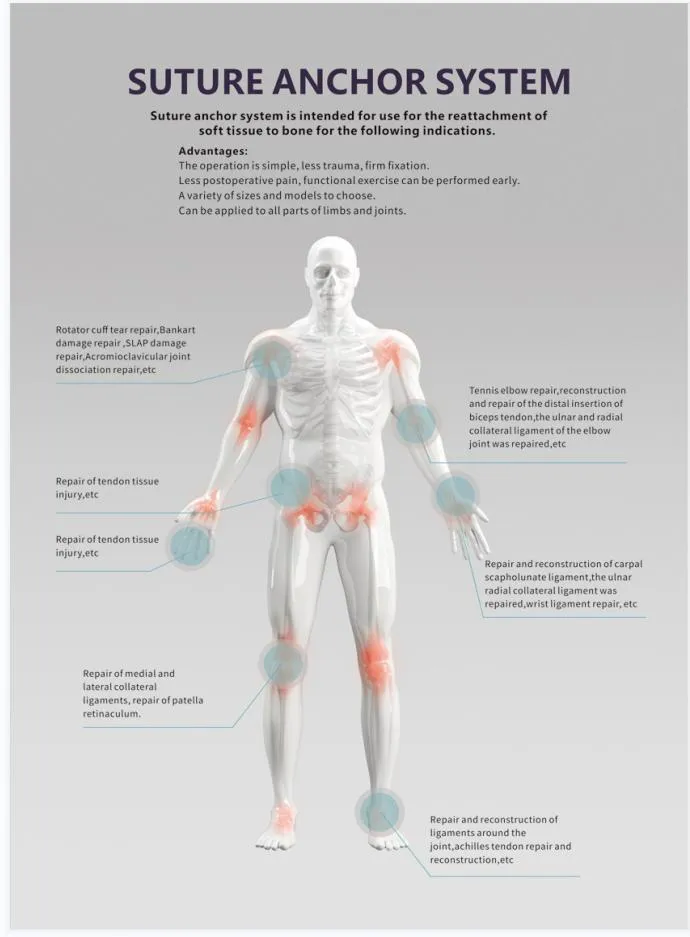
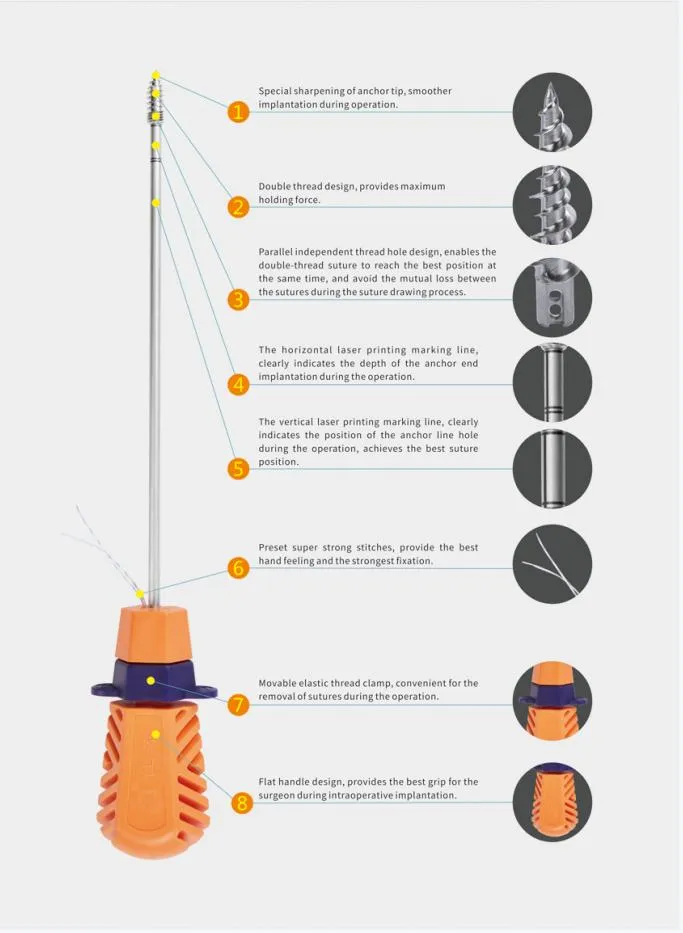
Suture Anchor System consists of anchor, nonabsorbable suture and inserter. The anchor is made of Ti6Al4V alloy, which meets the requirement of ISO5832-3:2016. Suture is made of ultra high molecular weight polyethylene (UHMWPE) material without coating, woven from several yarn, meeting ASTM F2848-17 standards.
The blue suture is made from white UHMWPE dyed with blue material (code 73.1015), approved by relevant authorities. The part of the inserter contacting the human body is high-quality stainless steel (ASTM F899-12b). The handle is ergonomically designed from polycarbonate and ABS material for precision control during surgery.








| Item | Value |
|---|---|
| Properties | Implant Materials & Artificial Organs |
| Model Number | WZ-MDT-S-01 |
| Instrument Classification | Class II |
| Material | Titanium Alloy (Ti6Al4V) |
| Dimensions | Diameter 1.8 x 6mm |
| Package | Sterile Packaging 1pc/package |
| Application | Orthopedic Surgery / Implantology |

 WEX Medical
WEX Medical