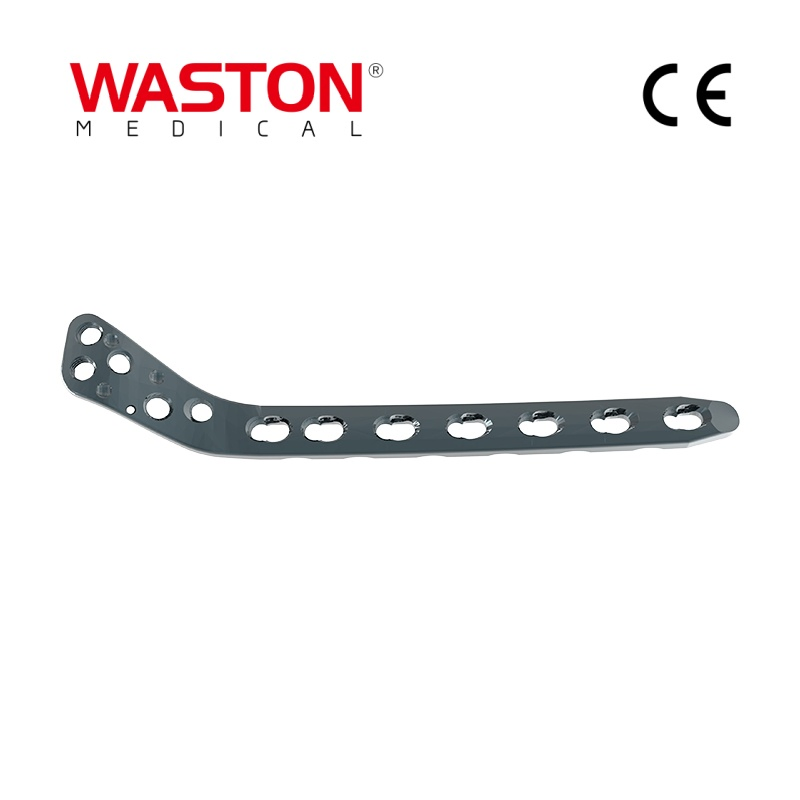














| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
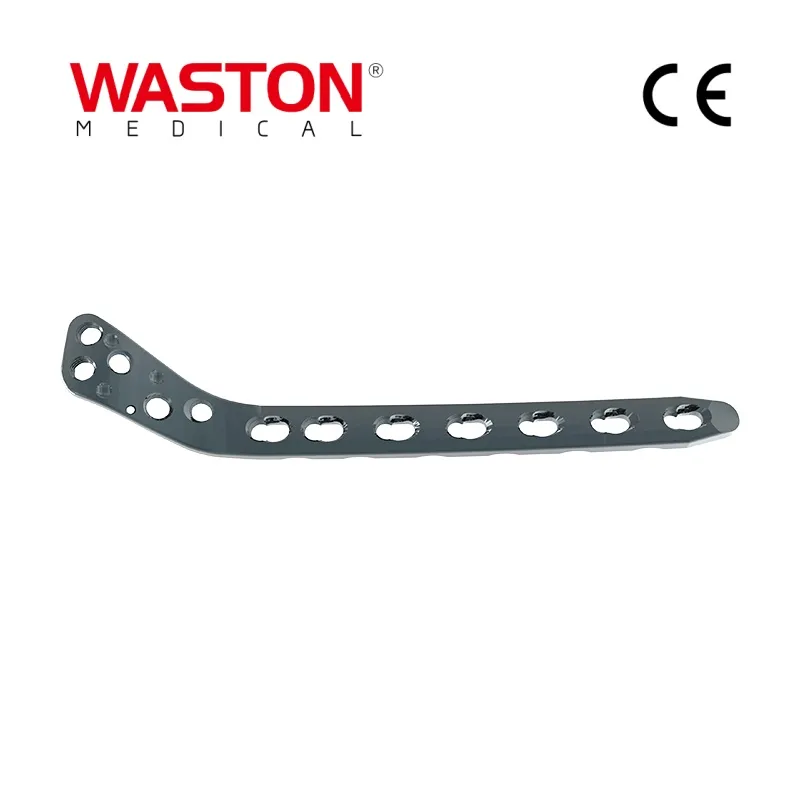
| PRODUCT CODE | L/R | HOLES | LENGTH (mm) |
|---|---|---|---|
| 10732-006 | L | 6 | 88 |
| 10732-106 | R | 6 | 88 |
| 10732-007 | L | 7 | 101 |
| 10732-107 | R | 7 | 101 |
| 10732-008 | L | 8 | 114 |
| 10732-108 | R | 8 | 114 |
| 10732-010 | L | 10 | 137 |



As a national high-tech enterprise, the proportion of R&D investment to total sales has been over 8% for consecutive years. We have launched several innovative products which have national invention patents and patents for utility model. We pioneer revolutionary innovation technologies including Rev Drill system, Three-Row-Stapler, Sternal Fixation and Rib plate.

Operating strictly according to the ISO13485 quality system, we adhere to medical device manufacturing quality management specifications. Our facilities include German-imported and American-imported machining centers and Japanese-imported automatic lathes, ensuring first-class quality. The testing center includes a biomechanical laboratory and a physical-chemical testing center for rigorous raw material and product control.
Products are exported to many countries, such as Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. We also serve as a major OEM supplier to large global players in the industry.



 WEX Medical
WEX Medical