
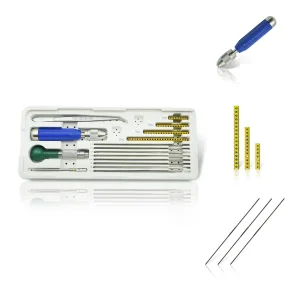
Veterinary Fixator Mini External Fixation System for Exotic Pet Surgery

Parts & Instruments of Cyclic Annular External Fixator External Fixation

Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set
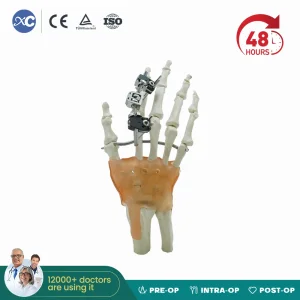
High Quality Mini Fragment External Fixator Orthopedic Medical Metacarpals External Fixation


The global orthopaedic implant market, particularly the Endobutton fixation sector, is witnessing an unprecedented surge. As sports-related injuries like ACL (Anterior Cruciate Ligament) tears become more prevalent among both professional athletes and the general population, the demand for reliable, biocompatible, and high-tensile fixation devices has skyrocketed.
Currently, the market is characterized by a shift towards minimally invasive surgery (MIS). Surgeons across Europe, North America, and the Asia-Pacific region are increasingly opting for cortical suspension devices, such as the Endobutton, due to their ability to provide superior graft-to-bone healing and simplified surgical workflows. This technology has become the "Gold Standard" for femoral fixation in ligament reconstruction.
Increasing adoption of biodegradable materials and 3D-printed titanium implants for better osseointegration.
Development of adjustable loop designs that allow for customized tensioning without complex knot-tying.
Global hospitals and clinics are prioritizing suppliers with CE and ISO certifications to ensure patient safety.
In the high-stakes world of orthopedic surgery, the margin for error is zero. Choosing from the top 10 suppliers ensures access to products that meet rigorous mechanical testing standards.
Top-tier exporters utilize medical-grade titanium (Ti-6Al-4V ELI), offering the perfect balance of strength, lightweight properties, and excellent biocompatibility, ensuring no adverse reactions in the patient's body.
Advanced manufacturing ensures that every Endobutton has smooth edges and exact dimensions to prevent suture abrasion, a critical factor in long-term surgical success.
Leading suppliers maintain CE, ISO 13485, and FDA approvals, facilitating smooth customs clearance and hospital audits in any country.
Changsha WEX Medical Instruments Co., Ltd. has experiences of more than two decades in areas of innovation and production of orthopaedic implants, such as bone plates, bone screws, interlocking nails, spine implants/internal fixation systems, and all kinds of instruments, which are branded by "WEX". All the implant products have certification of CE and ISO9001/ISO13485, so can reach different requirements of customers.
With a Quality Management System passed ISO 9001:2008 and ISO 13485:2003, we have bright and spacious factory workshops; more than hundred production equipments, such as advanced CNC machining centre machines, CNC sliding headstock auto-lathes, CNC milling machines, CNC lathing machines, upmarket ultrasonic cleaning equipments and etc; more than ten advanced and well-appointed testing equipment, such as Projectors, Tensile-testing machines, Durometers, Carbon and sulphur analyzers, Pitting LP detecting instruments and Fluorescent fault detectors and so on. The packaging process is conducted in clean workshop line with GMP standard.
At the beginning of company's establishment, we aim to design and produce implantable products professionally for our customers. Relying on the operational systems of development, design, production and marketing, which is formed by the orthopaedic experts in domestic large and medium-sized hospitals, also we completed the clinical insurance for our products.
We satisfy our customer's requirements with best quality and best service, based on our advanced production and processing technology. Because we insist on the idea of "Be professional for manufacturing high-quality products, be wholehearted for serving customers."


Ideal for high-volume ACL and PCL reconstruction surgeries where reliability and fast recovery are paramount for athletes.
Standardized fixation solutions that integrate seamlessly with existing surgical instrument sets and protocols.
Providing cost-effective, high-quality titanium implants to regions in Southeast Asia, Africa, and South America to improve local healthcare access.
The next generation of Endobuttons is moving toward suture materials that can be absorbed by the body after the ligament has successfully fused with the bone. This reduces the risk of long-term foreign body sensation and facilitates cleaner secondary procedures if needed. Suppliers are investing heavily in polymers like PEEK and PLDLA.
The industry is integrating with AI-driven surgical planning software. High-end exporters now provide 3D models of their Endobutton fixation systems, allowing surgeons to virtually "test" the fit and tensioning in a 3D reconstruction of the patient's knee before the first incision is made.
As global supply chains become more environmentally conscious, the "Top 10 Suppliers" are adopting green manufacturing processes. This includes reducing titanium waste during CNC milling and utilizing solar power in manufacturing plants, like our modern facility in Changsha.
China has evolved from a low-cost manufacturer to a global hub for high-precision medical technology. By partnering with a Chinese factory like WEX Medical, global distributors benefit from a robust supply chain, rapid prototyping capabilities, and a significant cost advantage without compromising on European quality standards (CE). This enables hospitals to manage their budgets more effectively while maintaining the highest level of patient care.