High-performance orthopedic implants designed for complex fracture management.
The industrial evolution and market dynamics of orthopedic trauma solutions.
The global trauma implants market is currently experiencing a transformative phase, driven by the increasing incidence of road traffic accidents, geriatric population growth, and technological breakthroughs in biocompatible materials. As a critical sub-segment of the orthopedic industry, trauma implants encompass a wide array of devices including plates, screws, intramedullary nails, and external fixators designed to stabilize bone fractures and facilitate biological healing.
Modern trauma surgery requires implants that are not only strong but also biologically "smart." The global market, led by major hubs in the USA, Europe, and China, is projected to reach several billion dollars by 2030. Exporters are no longer just selling hardware; they are providing comprehensive surgical systems. From titanium alloy locking plates to biodegradable polymer screws, the variety of products available today caters to specific anatomical requirements, ranging from the delicate maxillofacial region to heavy-load-bearing femoral structures.
Patient-specific implants are becoming the gold standard for complex reconstruction, allowing for a perfect anatomical fit.
The development of percutaneous instruments and smaller, stronger implants reduces recovery time and surgical risk.
PEEK (Polyether ether ketone) and advanced titanium alloys are replacing traditional steel for better elasticity matching bone properties.
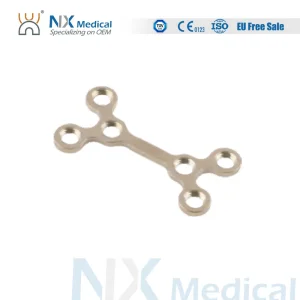
Our Maxillofacial Trauma Implants utilize the latest Variable Angle (VA) Locking Compression Plate technology. This allows surgeons to direct screws up to 15 degrees off the central axis, providing unmatched flexibility in complex fracture patterns. Manufactured from medical-grade Titanium and PEEK, these implants ensure biocompatibility and reduce the risk of post-operative complications.
Ideal for reconstructive surgery, our system is a testament to the high-tech manufacturing capability of modern Chinese factories, combining durability with the finesse required for facial trauma.
View Detailed SpecsProcurement departments in modern hospitals and clinics have shifted their focus from "lowest price" to "best value." The primary requirements for trauma implant exporters include:
Trauma implants are applied differently across various global regions. In developing economies, there is a massive demand for robust, cost-effective external fixators and intramedullary nails due to high rates of heavy industrial and transport injuries. Conversely, in aging populations like Japan and Western Europe, the focus is on fragility fracture management, requiring sophisticated locking plates that can provide stable fixation in osteoporotic bone.

Designed for the rigorous demands of femoral fracture repair, the PFNA (Proximal Femoral Nail Antirotation) System is a cornerstone of our trauma portfolio. It offers superior stability and ease of insertion, making it the preferred choice for surgeons dealing with intertrochanteric fractures.
The interlocking mechanism ensures that rotation is minimized, promoting faster bone union. As a leading trauma implants factory, we ensure that every nail is precision-turned on CNC sliding headstock lathes to meet sub-micron tolerances.
Technical BrochureLeading the way in orthopedic manufacturing excellence.
As a premier orthopedic trauma manufacturer based in China, Changsha WEX Medical Instruments Co., Ltd. represents the peak of industrial integration. Our advantages extend beyond simple manufacturing to encompass research, design, and a rigorous quality management system.
Utilizing high-end CNC machining centers and sliding headstock auto-lathes to produce complex geometries with absolute precision.
Testing includes tensile-testing, carbon/sulphur analysis, and fluorescent fault detection to ensure zero-defect exports.
Our products are developed in partnership with orthopedic experts from leading hospitals to ensure practical surgical utility.
Changsha WEX Medical Instruments Co., Ltd. has experiences of more than two decades in areas of innovation and production of orthopaedic implants, such as bone plates, bone screws, interlocking nails, spine implants/internal fixation systems, and all kinds of instruments, which are branded by "WEX". All the implant products have certification of CE and ISO9001/ISO13485, so can reach different requirements of customers.
With a Quality Management System passed ISO 9001:2008 and ISO 13485:2003, we have bright and spacious factory workshops; more than hundred production equipments, such as advanced CNC machining centre machines, CNC sliding headstock auto-lathes, CNC milling machines, CNC lathing machines, upmarket ultrasonic cleaning equipments and etc; more than ten advanced and well-appointed testing equipment, such as Projectors, Tensile-testing machines, Durometers, Carbon and sulphur analyzers, Pitting LP detecting instruments and Fluorescent fault detectors and so on. The packaging process is conducted in clean workshop line with GMP standard.
At the beginning of company's establishment, we aim to design and produce implantable products professionally for our customers. Relying on the operational systems of development, design, production and marketing, which is formed by the orthopaedic experts in domestic large and medium-sized hospitals, also we completed the clinical insurance for our products.
We satisfy our customer's requirements with best quality and best service, based on our advanced production and processing technology. Because we insist on the idea of "Be professional for manufacturing high-quality products, be wholehearted for serving customers."


Complete instrumentation and implant systems for all orthopedic specialties.