Manufacturing Capability
Established in 1958, the company is engaged in the development, manufacture and sales of orthopedic implants and instruments. As a director company of the Medical Apparatus Industry Association, the product line covers spine pedical screws, interlocking nails, locking plates/screws, trauma plates/screws, cannulated screws, external fixators, hip prosthesis and surgical instruments.
Advanced equipment including imported CNC, high precision digital control machines, and a 100,000 Grade axenic purification chamber contribute to the high quality of the products. The company was the first to pass ISO 9001 quality system certification. The products have won national silver awards and invention prizes.
Frequently Asked Questions
What are the primary products available?
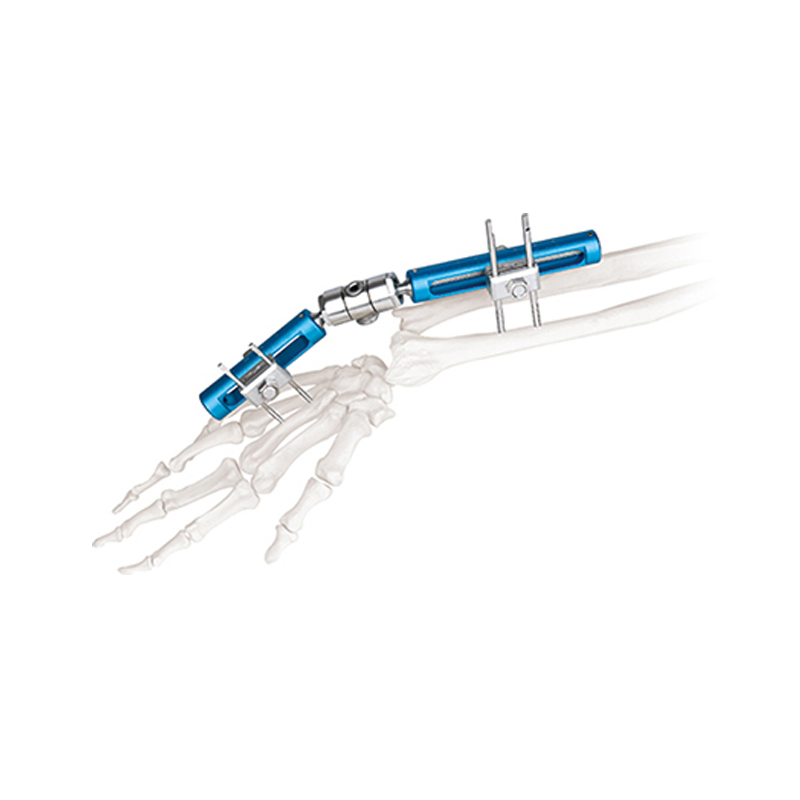
The main products include trauma plates, interlocking nails, spinal fixators, external fixators, and power tools for orthopedic surgery.
How is product quality guaranteed?
All products are produced according to EU CE standards. A pre-production sample is always evaluated before mass production, followed by a final inspection before shipment.
What materials are used in the external fixators?
High-quality Aluminum (AL) and Stainless Steel (SS) are primarily used to ensure durability and biocompatibility.
Is OEM/ODM service supported?
Yes, OEM and ODM orders are accepted to meet specific clinical and market requirements.
What are the accepted payment and delivery terms?
Delivery terms include FOB, CIF, EXW, and Express Delivery. Accepted payment methods include T/T, L/C, Western Union, and Cash in USD, EUR, or CNY.
What certifications do the medical devices hold?
The products hold CE and ISO13485 certifications, complying with international medical quality management systems.























 WEX Medical
WEX Medical